Polio
This article was first published in November 2017, and we updated the text in May 2024.
Polio is an infectious disease that can lead to the permanent paralysis of various body parts and can ultimately cause death by immobilizing the patient’s breathing muscles. It primarily affects children.
No cure exists for the symptoms, but in the 1950s effective vaccines were developed and have been used around the world since then. This allowed some richer countries to eliminate the disease in the 1960s and ’70s. But large outbreaks continued around the world. In the early 1980s, there were hundreds of thousands of cases globally each year1 and the disease was still prevalent in over a hundred countries.
As a response the "Global Polio Eradication Initiative" (GPEI) was founded in 1988 to fight the virus's spread and disease burden through a global vaccination campaign.
Since then, the world has made rapid progress against the disease. Two of the three types of wild poliovirus have been eradicated worldwide, and one remains.
See all interactive charts on polio ↓
Related topics
Other research and writing on polio on Our World in Data:
- The global fight against polio — how far have we come?
- New polio vaccines are key to preventing outbreaks and achieving eradication
- We need more testing to eradicate polio worldwide
- Estimation of the number of paralytic polio cases by region
Polio
Symptoms and transmission of polio
Polio, short for poliomyelitis, is an infectious disease that is caused and transmitted by a virus called the poliovirus.
Polio can cause paralysis and the disease is therefore also known as "infantile paralysis". The name poliomyelitis is derived from Greek and translates to gray (polios) marrow (myelon), which refers to the tissue in the center of the spinal cord, which when affected causes paralysis. Paralyzed limbs, such as arms or legs, waste away over time — this is why deformed legs are commonly associated with the disease.
The symptoms vary widely between patients. Most infections do not lead to any symptoms, but others suffer terribly and for some, it leads to death. Between 1 in 50 and 1 in 500 infections result in paralysis, among people who have not been vaccinated.2
The majority of infections (72%) do not lead to any symptoms. About a quarter of cases (24%) result in “abortive” poliomyelitis which leads to nonspecific symptoms for a few days, such as a fever or a cold, and 1–5% of cases lead to “non-paralytic aseptic meningitis”, in which the patient suffers from stiff limbs for up to 10 days.3
The poliovirus is found only among humans and is transmitted via the so-called fecal-oral route. In other words, polio is mostly transmitted by drinking water that has been contaminated by the feces of a person carrying the poliovirus.
The virus therefore spreads especially well in conditions of poor sanitation — for example, when people defecate in the open or do not filter their water before drinking it. The fact that the virus can only survive in humans (and no other animals) makes it possible to completely eradicate the disease from the world.
Polio is difficult to track in the population because it has a relatively long incubation period of up to 10 days and around three-quarters of infections do not cause symptoms, so the virus can spread for several months without being detected.
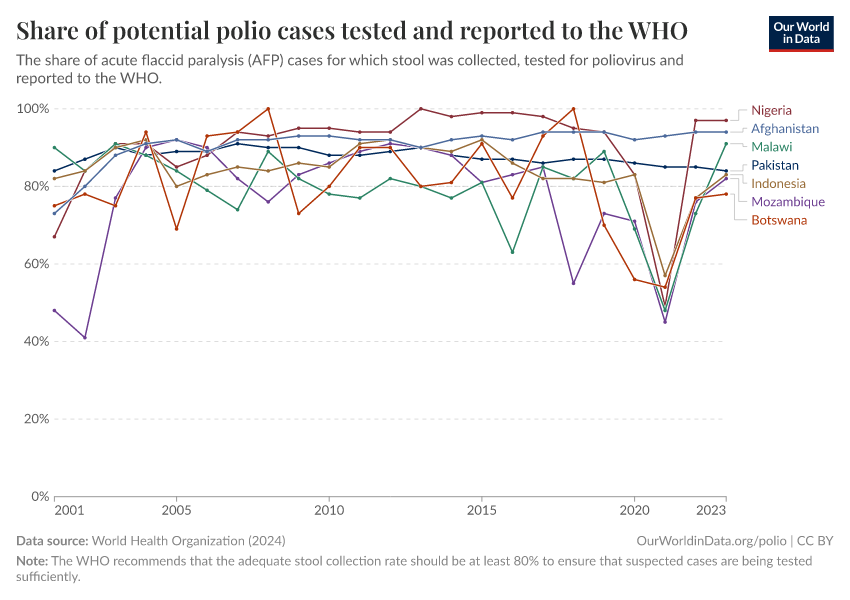
Monitoring has to focus on identifying patients that suffer from symptoms or rely on stool samples. Because any single cases that have been identified might suggest there are larger outbreaks, the WHO recommends that a single case of wild polio in a child should be treated as a public health emergency, in a country that was previously declared polio-free.4
Fighting polio's symptoms: The "Iron Lung'
Polio can lead to the death of infected patients if the paralysis immobilizes their breathing muscles, leading to suffocation.5 To prevent death by suffocation, Harvard professors Philip Drinker and Louis Agassiz Shaw invented the so called “iron lung”6 (shown in the picture) in 1928.
Infected patients would be placed in an air-tight tube — with their heads outside — and the machine would reduce the pressure inside the box, to induce inhalation, before returning to the normal outside pressure conditions, to induce exhalation.
Most patients would spend one to two weeks in an iron lung before their paralytic symptoms faded and they could breathe independently once again. If people experienced permanent paralysis, on the other hand, they would be bound to live inside the iron lung for years.
As it is often the case with innovations, the iron lung became only widespread when the price declined substantially. John Emerson managed to construct an iron lung at half the cost in 1931. In the meantime, Philip Drinker had tried to protect his invention with a patent and therefore filed a lawsuit against Emerson for using the original iron lung set-up. Emerson did not succumb, claiming that life-saving devices should be made available to the public at low cost rather than be used for private financial gains, and in the end, Drinker's lawsuit failed.8
The falling price of the Iron Lung enabled mass distribution and widespread use from the end of the 1930s onwards. The cost of one iron lung in the 1930s amounted to US-$1,500, which is the equivalent of approximately US-$26,000 in 2016 when accounting for inflation.9 The design quickly spread to Western Europe and was widely used for polio patients there, too.
Empirical View
Historical Perspective
The history of polio can be divided into three major phases:10
- The endemic phase from antiquity to the nineteenth century in which the disease occurred relatively rarely and did not result in many paralytic cases.
- The epidemic phase until the mid-20th century, during which the world saw large-scale outbreaks and increased geographic spread.
- The vaccine phase that followed the introduction of vaccines in 1955. In this phase, polio prevalence declined first in richer countries and over the last decades in poorer countries around the world.
The hope is that the world will see a fourth and final phase, in which polio is entirely eradicated from the world.
The origin of polio
Infectious diseases are generally believed to arise from the interplay of various developments such as human settlement in urban structures, crowding that causes poor hygiene, food shortages enhancing populations' morbidity, and the domestication of animals.
The exact origins of the disease are unknown, but based on the characteristics of the disease, epidemiologists can hypothesize how the disease has evolved and spread.11
Because the disease requires a human host and does not survive outside the human body for longer than one to two weeks, the disease could only have developed when humans started to settle in larger groups.
Various skeletons have been found with deformations similar to polio12 but the most widely-referenced indication of polio has been an Egyptian stele (pictured) depicting Doorkeeper Roma with one leg skinnier and deformed, both typical symptoms of paralytic polio.
The prevalence of polio in the past
Since then, evidence of occurrences of polio has been scarce. But, at different time points, records of polio-like diseases have appeared.
The poet Sir Walter Scott was infected with polio at the age of eighteen months in 1773 and the disease paralyzed his right leg for life. In his Treatise on the Diseases of Children in 1789 the London pediatrician Michael Underwood writes about a children's disease, possibly polio, that is a "debility of the lower extremities".14
Other outbreaks in the 19th century, which recorded a smaller number of cases, can be seen in the table taken from Smallman-Raynor & Cliff (2006).15
Polio-like outbreaks in the 19th century16
Year | Location | Cases (deaths) |
|---|---|---|
1808 | Göteborg, Sweden | 4 (-) |
1835 | Worksop, UK | 4 (-) |
1841 | Louisiana, USA | 10 (-) |
1868 | Modums, Norway | 14 (4) |
Polio in the epidemic phase
Until the 19th century, populations experienced relatively small outbreaks. This changed around the beginning of the 20th century. Major epidemics occurred in Norway and Sweden around 1905 and later also in the United States.17
Why did we see such large outbreaks of polio only in the 20th century? Or, in other words, why did the transition from the endemic to the epidemic phase take place?
The answer, again, lies with hygiene standards. As polio is transmitted via the fecal-oral route, the lack of flush toilets and the lack of safe drinking water meant that children in the past had usually been exposed to the poliovirus before their first birthday.
At such a young age, children still benefit from “passive immunity”, which is passed on from their mothers in the form of antibodies. These are proteins that identify the poliovirus as something foreign and therefore signal to the body that they should be eliminated by the immune system. Thereby, virtually all children would contract the poliovirus at a very young age.
In addition, while protected from developing the disease thanks to the maternal antibodies, children would also produce memory cells in response to the virus, which ensured long-term immunity against polio. The latter is important as the mother's cells have a half-life of only around a month, starting from the last day of breastfeeding.18
Once the maternal antibodies decrease sufficiently, children lose their passive immunity.
As hygiene standards improved, the average age at which children were first exposed to the poliovirus increased, which meant that maternal antibodies were no longer present to protect children from polio.
For example, during five epidemics in the US between 1907 and 1912, most reported cases occurred in one- to five-year-olds, whereas during the 1950s, the average age of contraction was 6 years, with "a substantial proportion of cases occurring among teenagers and young adults".19
Being exposed to the poliovirus after losing the protection from maternal antibodies meant that they were more likely to get polio, which increased the number of cases and deaths around the start of the 20th century.
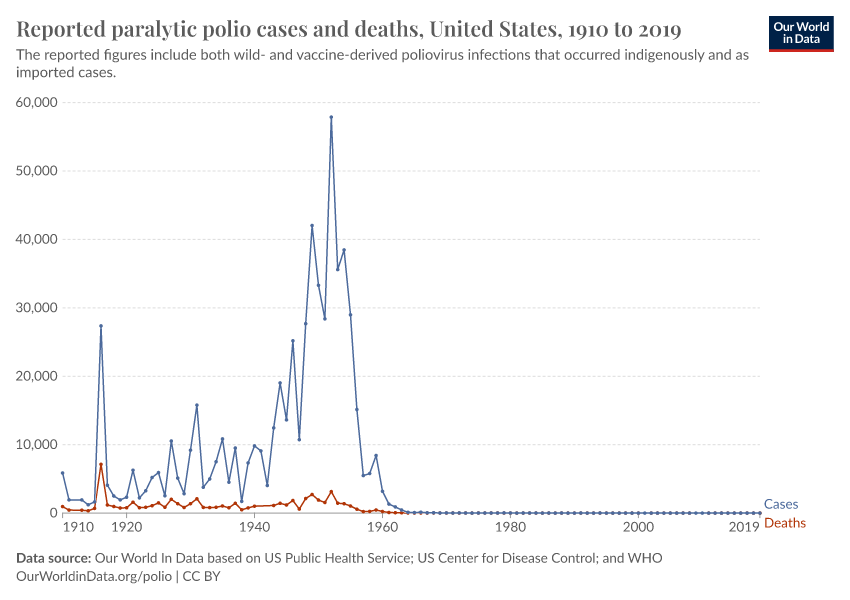
The history of polio in the US
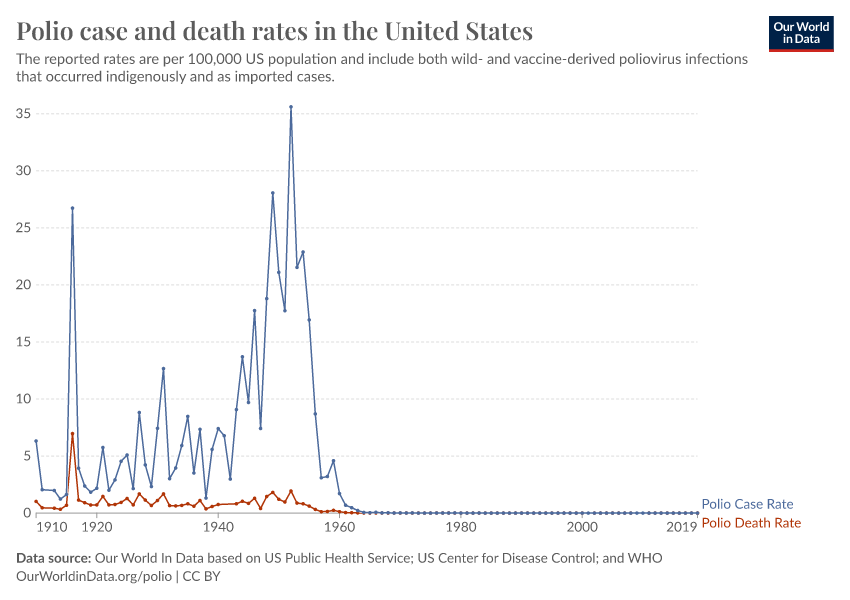
The chart here shows the annual absolute number of reported deaths and cases in the United States over the last century; the corresponding perspective on the rate of deaths and cases is shown in this visualization.
Big outbreaks happened frequently. In 1916 for example, the poliovirus infected more than 27,000 Americans and killed more than 7,000 people. At that time, the cause of the disease and how it spread were not yet known, so panicked New Yorkers shut down schools, public cinemas, and swimming pools. For a while, it was believed that cats or mosquitoes were spreading the virus, which led to the killing of more than 72,000 cats and the extensive spraying of the insecticide DDT, in a futile attempt to interrupt the transmission of the virus.20
Each of these large outbreaks came to an end because, like most viral diseases, the spread of the poliovirus in moderate climates like in the USA is seasonal and is mostly transmitted during the summer months.
By October 1916, enough New Yorkers had been infected and developed immunity in response so that in combination with the natural seasonal decline of the virus's spread the case numbers had already dramatically dropped and would not surge again.
This can be seen in the graph as the US recorded less than 5,000 cases in the following year. The second major outbreak in the USA in the 1950s, on the other hand, was largely contained by the successful development of polio vaccines that would hinder the transmission of the virus.
The vaccine against polio
The development of the polio vaccine
What changed the history of polio forever was the development of a vaccine against the disease.
US President Franklin D. Roosevelt himself had been diagnosed with polio at the age of 39 and subsequently bound to a wheelchair for the rest of his life. While this might have been a misdiagnosis in Roosevelt's case,21 his presidential influence was crucial in the set-up of the National Foundation for Infantile Paralysis. The non-profit organization soon became known as "The March of Dimes Foundation", referring to polio victims' inability to walk, and successfully collected a substantial amount of donations for vaccine research and its "Iron Lung" distribution program.
Years of research went into the effort to develop an effective vaccine.
The medical doctor and virologist Jonas Salk put forward a promising vaccine — known as the inactivated polio vaccine or the Salk vaccine — and in the spring of 1953, the foundation rolled out a large-scale trial for which 1.83 million children in 44 US states received either a placebo or the vaccine shot.22 Salk's supervisor Thomas Francis insisted on introducing a control group into the trial design, which was a step towards the development of randomized controlled trials in medicine.
The foundation was supported mainly by donations from the American people, who were collecting dimes, quarters, and dollars for decades in the hope of research finally uncovering a way to protect oneself against polio. Oshinsky (2005) even reports that the foundation received donations from two-thirds of the US population and a poll claims that more Americans knew about the field trials than about the president's full name (Dwight David Eisenhower).23
On April 12, 1955, the tenth anniversary of Roosevelt's death, Francis announced that Salk's vaccine was effective and potent in preventing polio. Within just two hours, the US Public Health Service issued a production license and the foundation prepared for a national immunization program. The conference had been live-broadcasted to physicians all over the country who had gathered in movie theaters to watch the announcement, millions of Americans received the news over the radio, spontaneously putting down their work in celebration of the news.24
At 10:30 PM of the same day, Thomas Francis and Jonas Salk gave a televised live broadcast interview in which Salk, when asked who owned the patents to the vaccine, famously answered "Well, the people I would say. There is no patent. Could you patent the sun?"25
His answer was in the spirit of the foundation having funded the vaccine research with donations from the American public and his conviction that life-saving technology should be for the benefit of society as a whole rather than for private financial gains.
Shortly afterward, Dr. Albert Sabin introduced a live polio vaccine that could be administered orally (in contrast with Salk's vaccine, which was given by injection), the oral polio vaccine (OPV).26
While Salk's vaccine only protected the central nervous system, Sabin's vaccine also protected the digestive tract and thereby prevented the spread of the wild poliovirus more effectively.
The easier administration also made vaccination efforts less expensive as it did not require trained health workers to provide injections. For these reasons, OPV has been used around the world and it is the vaccination that is responsible for the dramatic reduction in polio infections globally that we document below.
Polio prevalence in the developing world and the global spread of the polio vaccine
Dr. Sabin's Oral Poliovirus Vaccine (OPV) was tested on more than 100 million people in the Soviet Union before obtaining its license in 1961. Because its production costs were lower and the oral administration easier, OPV was and still is the predominant vaccination serum in many countries.
It only became apparent during the 1970s and 1980s, through several polio surveys in poorer countries that became known as "lameness surveys", that polio was as much a problem in developing countries as it was in Western Europe and the United States.
Bernier (1984)27 cites 46 of these "lameness surveys" in 24 countries, with a prevalence of paralytic polio ranging from less than one to 19 cases per 1,000 children. Modlin (2010)28 claims that these countries suffered from a higher prevalence of polio than the USA during its peak polio outbreaks.29
Mass campaigns in Brazil, Cuba, and Mexico proved the vaccine's effectiveness in different geographical areas but only with the foundation of the Global Polio Eradication Initiative at the World Health Assembly in 1988 did the polio vaccine find its place in many national routine immunization programs.
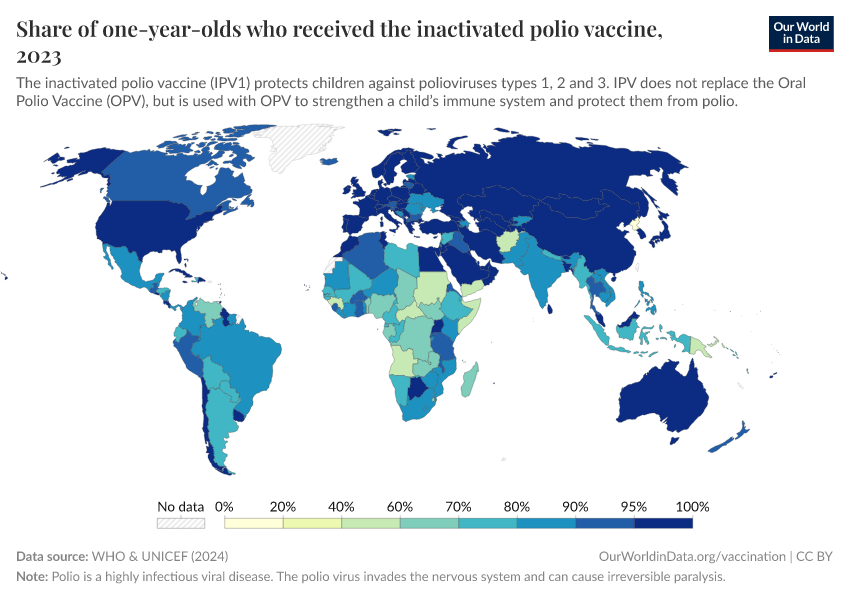
The visualization below shows how vaccine coverage against polio among one-year-olds has substantially risen around the world since 1980.
By switching to the chart view, you can see the change over time for each country and the world as a whole. Globally you can see that in 1980 only 22% of one-year-olds were vaccinated against polio. By the 2010s, over 80% were vaccinated.
Vaccine-induced polio
In rare cases, the altered live poliovirus that is used in the oral poliovirus vaccine (OPV) can mutate and regain its ability to attack the central nervous system ("neurovirulence"). This means that a small share of people who receive the OPV vaccine develop paralysis, which has the same symptoms as paralysis from the wild poliovirus.
There are two ways this can occur:
- Vaccine-associated paralytic poliomyelitis (VAPP). If the mutations to regain neurovirulence occur spontaneously — in a person who was recently vaccinated — it is called vaccine-associated paralytic poliomyelitis (VAPP). This is very rare: for every million doses of oral poliovirus vaccines, there have been between 0.09 and 25 cases of vaccine-associated paralytic poliomyelitis.30
- Vaccine-derived poliovirus (VDPV). If the mutations to regain neurovirulence occur over a longer period, it is known as vaccine derived poliovirus (VDPV). This can be identified by its genetic similarity to the genome of the virus used in the vaccine.
Vaccine-derived polioviruses
Let’s look into VDPVs in more detail. There are several types of VDPVs. They are defined by the GPEI as follows.31
- Immune deficiency associated vaccine derived poliovirus (iVDPVs) — these are VDPVs that come from people who have primary immunodeficiencies.
- Circulating vaccine derived poliovirus (cVDPVs) — these are VDPVs for which there is evidence that they have been transmitted between people in the community (i.e. they are found in multiple individuals who are not direct contacts; from an individual and an environmental sample; or from two environmental samples from different sites or at different times). These can spread in the community and cause new outbreaks.
- Ambiguous vaccine derived poliovirus (aVDPVs) — these are VDPVs for which there isn't evidence that they come from people with primary immunodeficiencies or have been transmitted between people in the community.
The risk of vaccine-derived polioviruses circulating in the population is generally very low, and the world now has new effective vaccines to contain them, called novel Oral Poliovirus Vaccines (nOPV).32
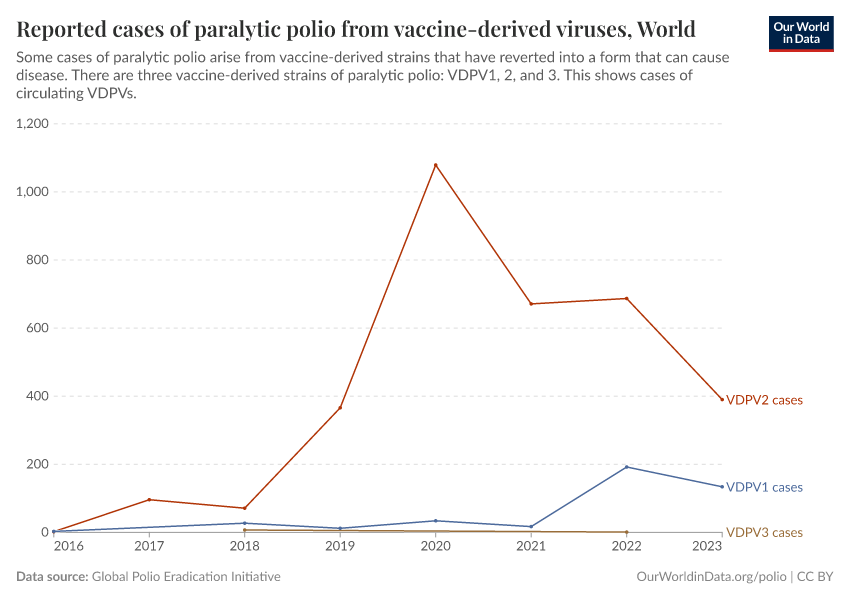
The vast majority of cases of cVDPVs come from the oral poliovirus vaccine against poliovirus serotype 2, rather than vaccines against other serotypes. You can see this in the chart.
In the vaccines against serotypes 1 and 3, many genetic mutations distinguish them from the wild poliovirus strains, which means it is extremely unlikely that they can revert into a form that can cause disease.
However, in the vaccine against serotype 2, fewer genetic mutations distinguish it from the wild poliovirus strain, meaning that in some rare cases, it can revert into a form that can cause disease.33
Although it may seem counterintuitive, cVDPV2 outbreaks are more likely in communities with lower coverage of vaccines for poliovirus serotype 2, such as OPV2 vaccines. This is because these communities have lower immune protection against poliovirus serotype 2, which some vaccine-derived viruses have reverted into.34
Although the wild poliovirus serotype 2 was eradicated — it was last reported worldwide in 1999 — cVDPV2 cases have occasionally occurred in under-vaccinated communities in the years that followed.
In the map, you can see the number of cases of all cVDPVs in each country.
How can vaccine-derived polioviruses be contained?
There are multiple ways to contain cases of cVDPVs.
In 2016, the Global Polio Eradication Initiative recommended in 2016 that countries switch the vaccines they used — from using an OPV that contains vaccines for all three serotypes (called a trivalent vaccine) to another OPV which contains vaccines for only serotype 1 and 3 (called a bivalent vaccine).35
Along with this, they also recommended countries use inactivated poliovirus vaccines (IPV) towards serotype 2 instead of the OPV, because it does not have the risk of mutating to regain neurovirulence.36 However, this comes with a trade-off: the OPV vaccines are much cheaper and easier to administer than the IPV vaccines, which typically require injections.37
Since 2019, all 126 countries that previously used OPV now use at least one dose of IPV.38
Since 2020, countries have had a new vaccine to use against serotype 2: the novel oral poliovirus vaccine (nOPV2). These new vaccines are much more genetically stable than the previous OPV2 vaccines, which makes it much less likely that they will revert and gain neurovirulence.32
Like the previous OPV vaccines, the nOPV vaccines are easy to administer as they are given orally, and they are being rolled out to countries with cases of VDPV2 to contain those outbreaks.39
Global decline of polio
The campaign to eradicate polio globally: the Global Polio Eradication Initiative (GPEI)
In 1988, the World Health Assembly — the governing body of the World Health Organization (WHO) — launched the Global Polio Eradication Initiative (GPEI) which was tasked with eradicating the disease globally by the year 2000.40 The eradication of the disease in just 12 years was an ambitious plan, since polio was endemic in 125 countries of the world in 1988.
The GPEI was set up as a public-private-partnership and today brings together several organizations, among which are the WHO, UNICEF, the US Center for Disease Control and Prevention (CDC), Rotary International and the Bill and Melinda Gates Foundation.41
They had achieved that by way of routine immunization programs which entails the basic schedule of each infant receiving three Oral Polio Vaccines (OPV) before they reached the age of one.
Since its inauguration in 1988, the GPEI has offered support for these routine immunization programs to governments. But in addition to these the GPEI also ran:
- National Immunization Days (NIDs), on which children receive two doses of OPV 4-8 weeks apart regardless of their immunization history,
- outbreak response immunization programs, for which all children below the age of five years in the vicinity of a detected case of paralytic polio receive one OPV dose, and
- mopping-up immunization programs, during which under-five-year-olds living in outbreak-prone areas are visited in their homes and receive two OPV doses one month apart.
Even though the GPEI has not yet reached the goal of eradicating polio, it has been successful in reducing the prevalence of polio around the world: reported polio cases have been reduced greatly and two of three wild poliovirus serotypes have already been eradicated.
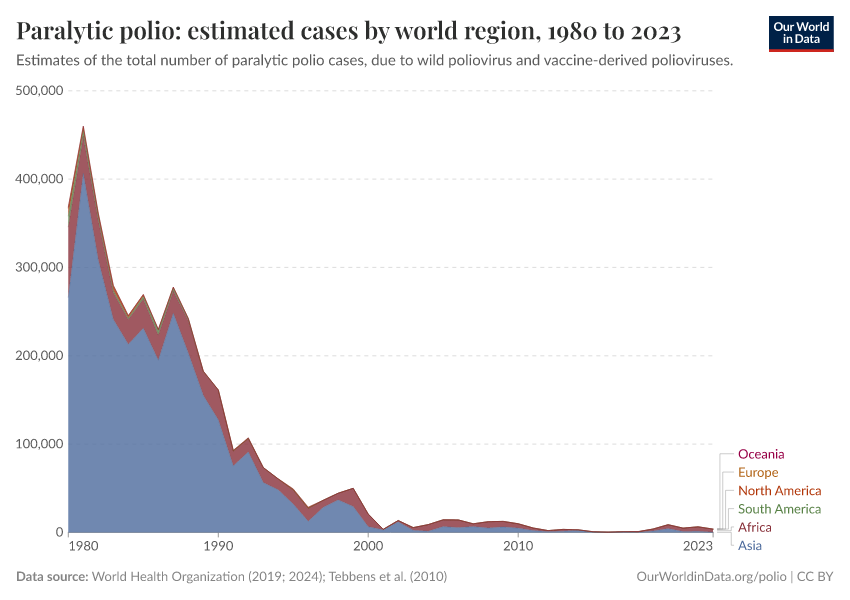
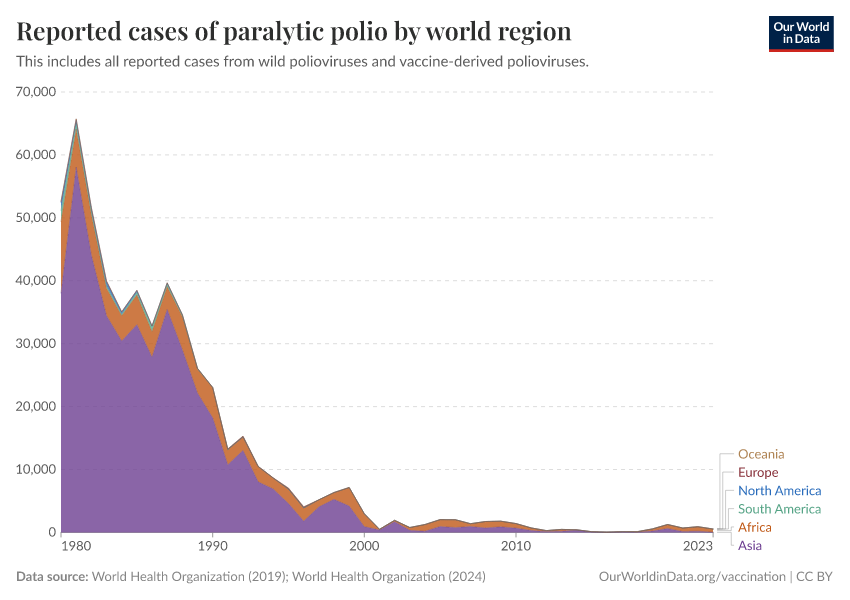
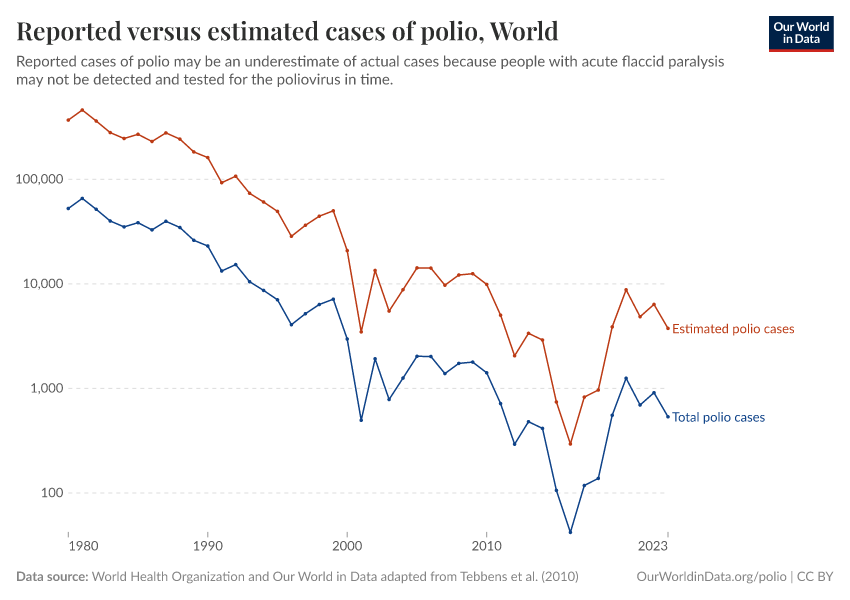
The number of estimated polio cases by world region
The interactive visualization highlights the global decline in the number of paralytic polio cases from 1980 onwards. In the early 1980s, an estimated 300,000 to 400,000 people suffered from paralytic polio cases every year. In 2020 there were 1873 paralytic polio cases. In the 1980s the world saw many more paralytic polio cases every week than today in an entire year.
The cases are shown for each of the six WHO world regions and you can change the view from absolute to relative numbers of polio cases by clicking on “Relative” in the chart. In the 1980s between 50% and 75% of all estimated cases occurred in the South-East Asia region, this region has not recorded a single case after 2011 and was certified to be polio-free in 2014.
This data shows the estimated total number of paralytic polio cases, which is adjusted from the number of reported paralytic polio cases.
Our estimations of the total number follow the methodology by Tebbens et al. (2010)42 who estimate the degree of underreporting — especially in earlier periods — of polio is and then adjust the number of paralytic polio cases to arrive at the total number of estimated cases.
Read more on how we adapted their method to apply correction factors here:
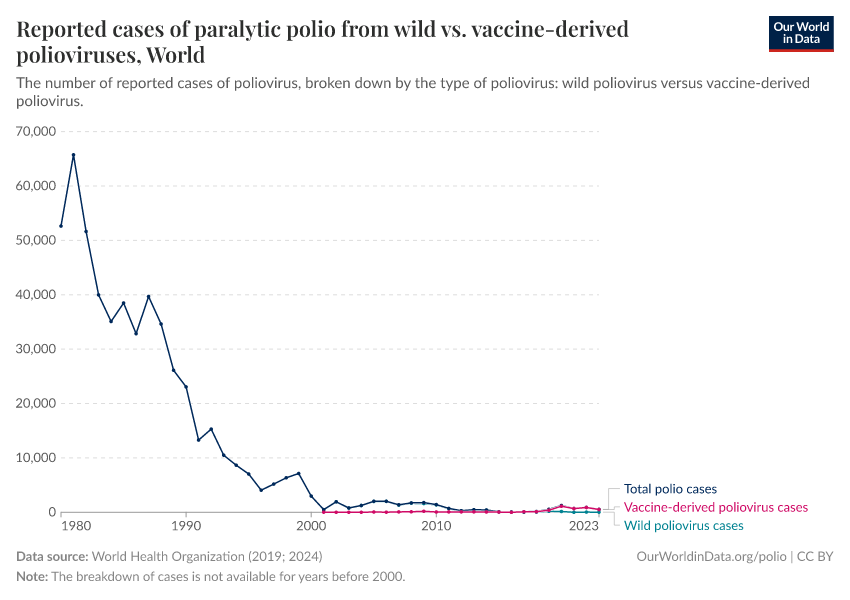
The total number of reported polio cases
The visualization documents the number of reported polio cases by country from 1980 onwards. The number of reported cases is an underestimate of actual polio cases, because some cases go undetected or unreported.
Reported polio cases include endemic, imported, and vaccine-derived cases.
For instance, the United States, which eliminated polio in 1978, still recorded some polio cases in the 1980s because imported cases were included.
This is also the case for countries that only recently achieved their polio-free status, such as India and Nigeria. Although Nigeria has been certified polio-free, this status refers to the wild poliovirus specifically. As you can see, several countries continue to report polio cases in 2021, which is due to vaccine-derived polioviruses.
Cases of polio have fallen dramatically over time. In 1980, there were over 50,000 reported cases of polio worldwide. But in 2021, this number was down to 649.
Country by country: The prevalence of polio
The interactive map presents the number of polio cases per 1 million inhabitants of each country, to account for differences in the population size and make comparisons between countries more meaningful.
You can press play in the bottom left corner to show the change over time. It can be clearly seen how the WHO regions achieved their polio-free status, the Americas for example were certified in 1994.
By clicking on any country you can see the change over time of the polio rate in that country.
In 2017 the wild poliovirus is only endemic in a few countries, shown in red on the map.
The map displays the year of the last recorded case of polio for each country and each decade is color-coded. You can see that the Americas were the first world region to be certified polio-free in 1994.
WHO-certified polio-free world regions and countries
The WHO certifies world regions as polio-free, rather than individual countries. The considered world regions are the six WHO world regions: Africa, Americas, Eastern Mediterranean, Europe, South-East Asia, and Western Pacific.
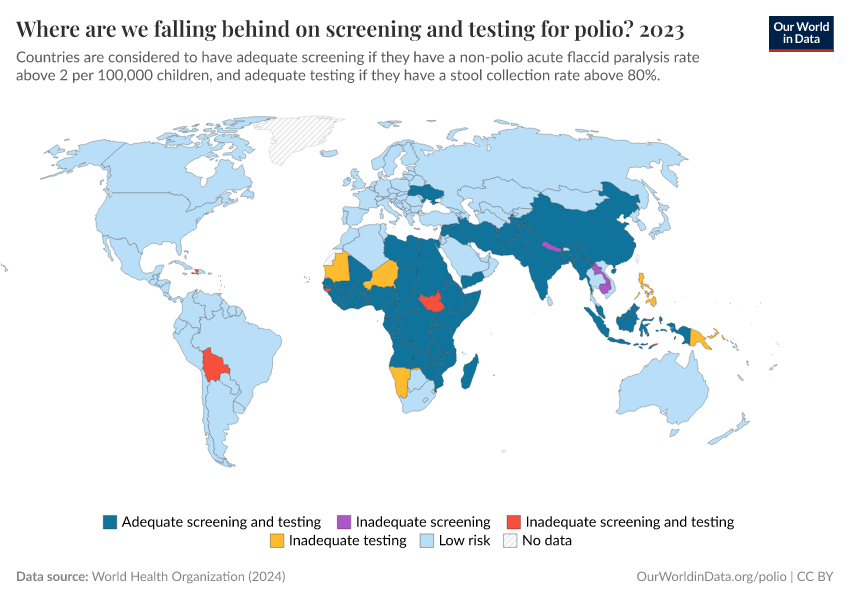
To be certified polio-free, a WHO region needs to (i) record no wild indigenous polio case for at least three years, (ii) have a reliable surveillance system in place, and (iii) prove its capacity to detect and respond to imported polio cases.43
The interactive map shows for each country when the last case of endemic paralytic polio was recorded. The map also shows when the four polio-free WHO regions achieved this status, three years after the last country in that WHO region recorded the last endemic polio case.
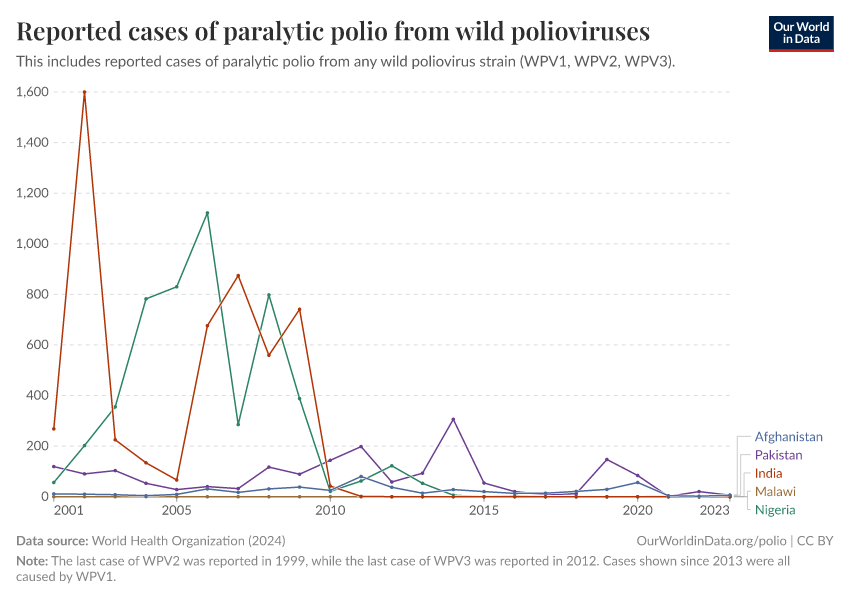
The number of reported polio cases from wild polioviruses
The visualization documents the number of reported polio cases from wild poliovirus (WPVs) by country. The number of reported cases is an underestimate of actual polio cases, because some cases go undetected or unreported.
Wild polioviruses refer to the polioviruses that have been historically endemic in many countries, and excludes vaccine-derived polioviruses. You can explore this data for other countries by clicking the “Edit countries and regions” button.
In 2001, 14 countries reported cases of wild polioviruses. By 2021 there were only three countries where wild poliovirus cases were recorded: Afghanistan, Pakistan, and Malawi.
Until recently, there were three strains of wild polioviruses. Poliovirus serotypes 2 and 3 have both been eradicated globally. The last case of wild poliovirus serotype 2 was seen in 1999 in India, while the last case of wild poliovirus serotype 3 was seen in 2012 in Nigeria. That means they were declared globally eradicated by the WHO in 2015 and 2019 respectively.
All reported cases since 2013 from wild polioviruses have been caused by wild poliovirus serotype 1.
The costs and benefits of eradicating polio
Health benefits
It is estimated that the Global Polio Eradication Initiative led to the prevention of 2.17 million cases of paralytic polio between 1988 (when the GPEI began) and 2018.44
The chart visualizes the benefit of the eradication campaign by comparing the actual cases with an alternative history in which the GPEI did not exist.
The red part of the chart is based on a model that estimates the number of paralytic polio cases without the eradication campaign, for the 105 countries that received support from the GPEI. Without the eradication campaign, this study assumed that the coverage rates of polio vaccines would match the rates of other routine vaccines in countries during the same time period.
The benefits of this global health campaign are not limited to polio itself. In addition to preventing paralytic polio, many children around the world have received other health benefits that were made available to them as part of the polio immunization campaigns. These are called supplementary immunization activities (SIAs), where other vaccinations and nutritional supplements have been distributed.45
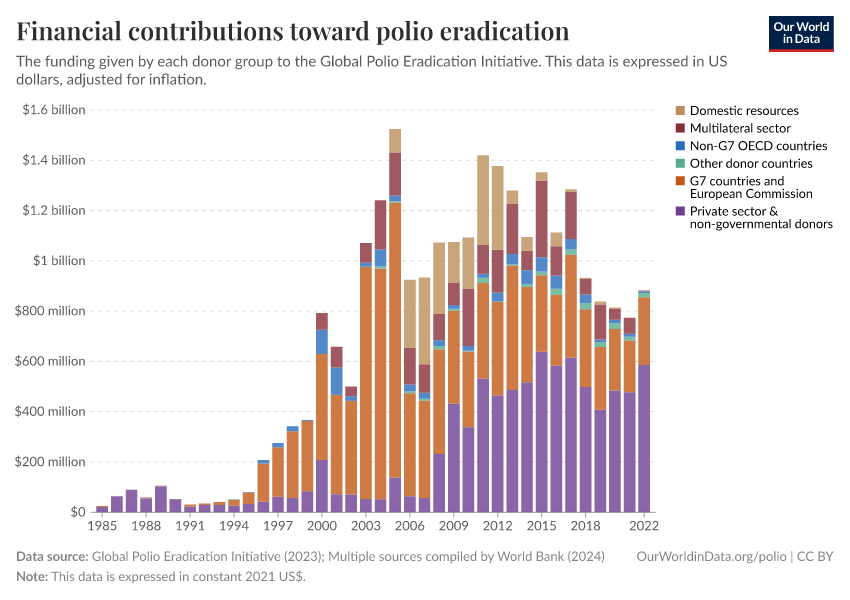
Economic costs of the Global Polio Eradication Initiative
The GPEI has received funding towards polio eradication from different sectors, including domestic resources, G7 countries and the European Commission, the private sector and non-governmental donors, as the chart below shows.
The large majority was only received from 2000 onwards. This funding contributed to additional polio eradication efforts in the form of National Immunization Days (NIDs), outbreak response immunization and mopping-up immunization.
Current expenditure on polio eradication and the Endgame Strategic Plan
In 2013, the GPEI launched an ambitious five-year plan to fully eradicate polio called Polio Eradication & Endgame Strategic Plan 2013-2018 which would cost US-$5.5 billion (in 2015 it was increased by a year and extended to US-$7 billion), also see the section on the benefits of eradication rather than reduction for more information.
Even though polio was endemic in only Nigeria, Pakistan, and Afghanistan in 2013, it proved especially difficult to monitor the virus and to reach every child for immunization in these contexts which makes the endgame strategy so expensive in comparison to the GPEI's previous budget.
That's why, in 2016, US-$536 million of the total GPEI budget of US-$925 million were spent in these three countries.46
The map indicates which countries received support from the GPEI in 2016. Only six of these fund their immunization efforts partially themselves.
The cost of the GPEI in a comparative perspective
To put these numbers into perspective, global malaria financing amounted to US-$2.9 billion in 2015 which was more than twice as large as the GPEI's budget of US-$1.39 billion one year later in 2016.
Or, to give a second comparison, in 2016, the US government spent US-$1,116 billion on major health care projects, a budget that is more than 800 times larger than the GPEI's global spending on polio in the same year.
Benefits of eradication rather than reduction
In 2013, the GPEI implemented “The Polio Eradication and Endgame Strategic Plan 2013-2018”, costing an additional $5.5 billion in addition to the already $9 billion spent by the organization since its implementation in 1988.
The initiative hopes to especially finance the last stretch of vaccination campaigns in the countries where polio is still endemic, keep the remaining countries polio-free, and closely monitor the occurrence for at least three years after the last reported case.
Even though the additional financial needs seem very high, the GPEI argues that eradication is the most cost-effective strategy, by illustrating that the long-term costs of controlling rather than eradicating will be substantially higher.
The benefits of eradicating polio extend beyond the health domain of having fewer people suffering from paralytic polio. In the economic domain, fewer polio patients translate into lower healthcare costs. Furthermore, once the virus has been eradicated, the world can stop producing and administering the polio vaccine as well as surveilling paralytic diseases suspected to be polio.
The GPEI argues that the discontinuation of these costly activities will therefore result in extensive economic gains from eradication as well. It is very difficult to accurately estimate these economic gains as it requires assumptions on for example the marginal economic value of a healthy person over a paralyzed polio patient or until what year you calculate these gains for. Tebbens et al. (2010)42 have attempted such a modeling exercise and arrived at a net benefit of US-$40-50 billion — when comparing the GPEI against just national routine immunization — in the time horizon between 1988 to 2035.
Such a cost-benefit analysis is made even more difficult by having to extrapolate the actual case counts from reported incidence figures, which is explained in more detail in our article here.
Key Charts on Polio
See all charts on this topicFeatured Data on Polio
Endnotes
Badizadegan, K., Kalkowska, D. A., & Thompson, K. M. (2022). Polio by the Numbers–A Global Perspective. The Journal of Infectious Diseases.
Nathanson, N., & Martin, J. R. (1979). The epidemiology of poliomyelitis: enigmas surrounding its appearance, epidemicity, and disappearance. American Journal of Epidemiology, 110(6), 672-692.
See "The Pinkbook" on poliomyelitis by the US Center for Disease Control and Prevention (CDC) here.
As a response, several million children under five years old are recommended to receive the vaccination.
See: Grassly, N. (2013). The final stages of the global eradication of poliomyelitis. Philosophical Transactions Of The Royal Society B: Biological Sciences, 368(1623), 20120140-20120140. Freely available online here.
As explained in the WHO's Factsheet on Polio.
For an introduction to the iron lung, the National Museum of American History has a good summary here.
Photo by Joe Clark. Repository: National Museum of Health and Medicine. Available online.
Here, you can read a condensed version from an article in The Nation, 1 March 1933, by Stewart Grimson, detailing the developments of the patent fight between Drinker and Emerson.
The amount of $1,500 stems from the National Museum of American History's summary. That sum of 1939 US-$ was then multiplied by the annualized inflation rate in the US from 1939 to 2016 and taken to the power of 77 (the number of years from 1939 to 2016) to arrive at 2016 US-$25,919.17 as the equivalent cost of an iron lung today).
Page 61 of Smallman-Raynor, M., & Cliff, A. (2006). Poliomyelitis (1st ed.). Oxford: Oxford University Press. Parts are available on Google Books.
The interested reader should refer to for example Eichner, M., Hadeler, K., & Dietz, K. (1996). Stochastic models for the Eradication of Poliomyelitis: Minimum Population Size for Polio Virus Persistence. In V. Isham & G. Medley, Models for Infectious Diseases: Their Structure and Relation to Data (pp. 315-330). Cambridge: Cambridge University Press. Partly available online on Google Books.
Their mid-point estimate of the size of a human community necessary for poliovirus to survive is 250,000 which would make the earliest surviving infections possible in Egypt (3,000 BC) or Sumeria, today's southern Iraq (2,700 BC).
Some of these paleopathologists have found skeletons with typical polio marks stemming from:
4000-2400BC Sussex, England (Wells, C. (1965). Bones, Bodies and Disease. Evidence of disease and abnormality in early man. London: Frederick A. Praeger.)
2000-500BC Norfolk, England (Roberts, C., & Manchester, K. (1995). The Archeology of Disease (2nd ed.). Stroud: Alan Sutton.)
3700BC Deshane, Egypt (Mitchell, J.K. (1900). Study of a Mummy Affected With Anterior Poliomyelitis. Transactions, 1. US: Transactions of the Association of American Physicians.)
1225BC Egypt (Pharaoh Siptah) (Nunn, J. (1996). Ancient Egyptian Medicine. London: University of Oklahoma Press.)
The picture is available on Wikimedia Commons.
Lockhart, J. (1837). Memoirs of the life of Sir Walter Scott. Edinburgh: Robert Cadell. Available online: https://archive.org/details/memoirslifesirw85lockgoog. and Underwood, M. (1789). Treatise on the Diseases of Children. Two Volumes. London: J. Matthews. Available online: https://archive.org/details/2575056R.nlm.nih.gov.
Their book is called Poliomyelitis (1st ed.). Oxford: Oxford University Press. Parts are available on Google Books.
The table is taken from Smallman-Raynor, M., & Cliff, A. (2006). Poliomyelitis (1st ed.). Oxford: Oxford University Press. Parts are available on Google Books.
It is based on
Holt, L. E., & Bartlett, F. H. (1908). The Epidemiology Of Acute Poliomyeitis. The American Journal of the Medical Sciences, 135(5), 647-661.
Batten, F. E. (1911). The epidemiology of poliomyelitis. Available online here.
Low, R. B. (1917). Forty-fifth annual report of the Local Government Board, 1915–16. Supplement containing the Report of the Medical Officer for 1915–16.
Lavinder, C., Freeman, A., & Frost, W. (1918). Epidemiologic Studies of poliomyelitis in New York City and the northeastern United States during the year 1916. Washington: Issue 91 of Public Health Bulletin (U.S. Government Printing Office).
A first large one happened in 1916 and a particularly large outbreak with 57,000 reported cases happened in the United States in 1952.
On page 3 of Robertson, S. (2017). The Immunological Basis for Immunization Series, Module 6: Poliomyelitis. Geneva: World Health Organization. Available online http://www.who.int/ihr/polio1993en.pdf.
Hall, W., Nathanson, N., & Langmuir, A. (1957). The Age Distribution of Poliomyelitis in the United States in 1955. American Journal Of Hygiene, 66(2), 214-34.
This trend was also observed across Scandinavia and Northern Europe, see for example Burnet, F. (1940). The epidemiology of poliomyelitis with special reference to the Victorian epidemic of 1937-1938. Medical Journal Of Australia, 1, 325-336.
Read a more detailed description of the 1916 outbreak in New York City in this article, written by Merelli, A. (2017). 100 years ago, New York City declared war against polio and killed 72,000 cats (and 8,000 dogs). Quartz. Retrieved from https://qz.com/787385/the-history-of-polio-in-new-york-includes-unnecessarily-killing-72000-cats-and-8000-dogs/.
Researchers recently re-evaluated the president's symptoms from his historical medical records and questioned whether he actually contracted polio or rather suffered from an auto-immune disease called Guillain-Barré syndrome (GBS). Goldman, A. S., Schmalstieg, E. J., Freeman Jr, D. H., Goldman, D. A., & Schmalstieg Jr, F. C. (2003). What was the cause of Franklin Delano Roosevelt's paralytic illness?. Journal of Medical Biography, 11(4), 232-240. It can be freely accessed online here.
Page 435 of Smallman-Raynor, M., & Cliff, A. (2006). Poliomyelitis (1st ed.). Oxford: Oxford University Press. Parts are available on Google Books.
Page 188 in Oshinsky, D. (2005). Polio: An American Story (1st ed.). New York: Oxford University Press. Parts are available on Google Books.
Illustrated in this article: King, G. (2013). Salk, Sabin and the Race Against Polio. Smithsonian. Retrieved from https://www.smithsonianmag.com/history/salk-sabin-and-the-race-against-polio-169813703/.
Oshinsky, D. (2005). Polio: An American Story (1st ed.). New York: Oxford University Press. Parts are available on Google Books.
For more information see the University of Virginia's overview of the development of the iron lung and polio vaccine which is available here.
Bernier, R. (1984). Some Observations on Poliomyelitis Lameness Surveys. Clinical Infectious Diseases, 6 (Supplement_2), S371-S375.
Modlin, J. (2010). The Bumpy Road to Polio Eradication. New England Journal Of Medicine, 362(25), 2346-2349. Retrieved from http://www.nejm.org/doi/full/10.1056/NEJMp1005405.
It is unfortunately not comparable to our graph of the United States' rate of paralytic polio cases, as it depicts the number of cases per population and not per children.
Some countries such as Romania, Belarus and Hungary have seen higher than average rates of VAPP in the population. High rates of VAPP in those countries have been linked to the use of intramuscular injections of antibiotics to treat other conditions, which led to a syndrome called "provocation polio." In other countries, the rates of VAPP have been far lower — with Brazil at the lowest end, having only 0.09 cases per million doses.
Platt, L. R., Estívariz, C. F., & Sutter, R. W. (2014). Vaccine-associated paralytic poliomyelitis: a review of the epidemiology and estimation of the global burden. The Journal of infectious diseases, 210(suppl_1), S380-S389.
Classification and reporting of vaccine-derived polioviruses (VDPV). (2016). Global Polio Eradication Initiative. https://polioeradication.org/wp-content/uploads/2016/09/Reporting-and-Classification-of-VDPVs_Aug2016_EN.pdf
Sáez-Llorens, X., Bandyopadhyay, A. S., Gast, C., Leon, T., DeAntonio, R., Jimeno, J., Caballero, M. I., Aguirre, G., Oberste, M. S., Weldon, W. C., Konopka-Anstadt, J. L., Modlin, J., Bachtiar, N. S., Fix, A., Konz, J., Clemens, R., Costa Clemens, S. A., & Rüttimann, R. (2021). Safety and immunogenicity of two novel type 2 oral poliovirus vaccine candidates compared with a monovalent type 2 oral poliovirus vaccine in children and infants: two clinical trials. Lancet (London, England), 397(10268), 27–38. https://doi.org/10.1016/S0140-6736(20)32540-X
Savolainen-Kopra, C., & Blomqvist, S. (2010). Mechanisms of genetic variation in polioviruses: Mechanisms of genetic variation in polioviruses. Reviews in Medical Virology, 20(6), 358–371. https://doi.org/10.1002/rmv.663
Jorgensen, D., Pons-Salort, M., Shaw, A. G., & Grassly, N. C. (2020). The role of genetic sequencing and analysis in the polio eradication programme. Virus Evolution, 6(2), veaa040. https://doi.org/10.1093/ve/veaa040
Yeh, M. T., Bujaki, E., Dolan, P. T., Smith, M., Wahid, R., Konz, J., Weiner, A. J., Bandyopadhyay, A. S., Van Damme, P., De Coster, I., Revets, H., Macadam, A., & Andino, R. (2020). Engineering the Live-Attenuated Polio Vaccine to Prevent Reversion to Virulence. Cell host & microbe, 27(5), 736–751.e8. https://doi.org/10.1016/j.chom.2020.04.003
Macklin, G. R., O’Reilly, K. M., Grassly, N. C., Edmunds, W. J., Mach, O., Santhana Gopala Krishnan, R., ... & Sutter, R. W. (2020). Evolving epidemiology of poliovirus serotype 2 following withdrawal of the serotype 2 oral poliovirus vaccine. Science, 368(6489), 401-405.
Global Polio Eradication Initiative. (2016, August 4). Global synchronisation and the switch. News Stories. https://polioeradication.org/news-post/global-synchronisation-and-the-switch/
Ramirez Gonzalez, A., Farrell, M., Menning, L., Garon, J., Everts, H., Hampton, L. M., ... & Patel, M. (2017). Implementing the synchronized global switch from trivalent to bivalent oral polio vaccines—lessons learned from the global perspective. The journal of infectious diseases, 216(suppl_1), S183-S192.
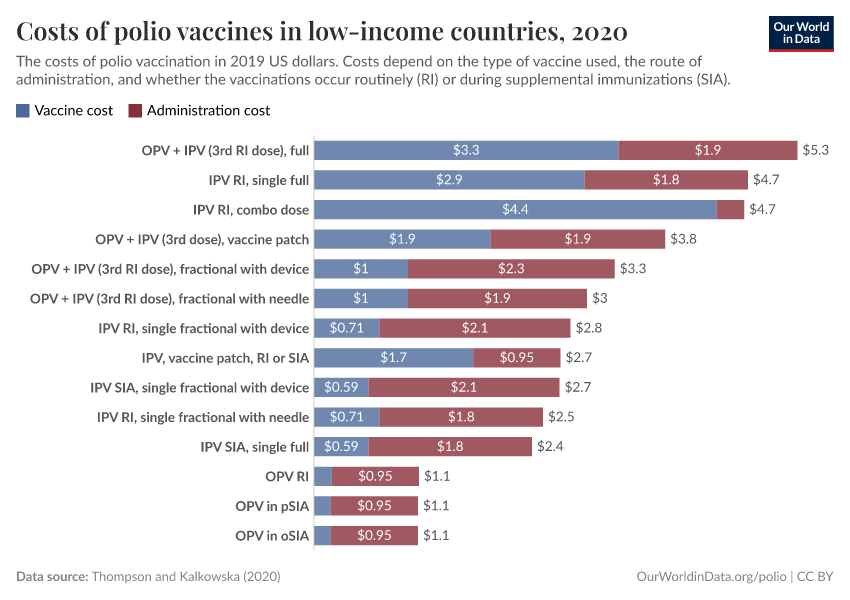
Thompson, K. M., & Kalkowska, D. A. (2021). Potential Future Use, Costs, and Value of Poliovirus Vaccines. Risk Analysis, 41(2), 349–363. https://doi.org/10.1111/risa.13557
Global Polio Eradication Initiative. (2019, September 5). Inactivated Polio Vaccine now introduced worldwide. News Stories. https://polioeradication.org/news-post/inactivated-polio-vaccine-now-introduced-worldwide/
Global Polio Eradication Initiative. (2021, October 10). Independent experts advise move to next use phase for novel oral polio vaccine type 2. News Stories. https://polioeradication.org/news-post/independent-experts-advise-transition-to-next-use-phase-for-novel-oral-polio-vaccine-type-2-nopv2/
This can be found in: World Health Assembly. Global eradication of poliomyelitis by the year 2000 (resolution 41.28). Geneva: World Health Organization; 1988.
Other partners include the United Nations Foundation and other private foundations, the World Bank, national governments, the European Commission, NGOs such as the International Red Cross and Red Crescent societies for example, private companies and 20 million volunteers. For more information on the GPEI's partners and donors, this is their website.
Tebbens, R., Pallansch, M., Cochi, S., Wassilak, S., Linkins, J., & Sutter, R. et al. (2010). Economic analysis of the global polio eradication initiative. Vaccine, 29(2), 334-343. Available online here.
As outlined in the WHO guidelines, for example here.
Thompson, K. M., & Kalkowska, D. A. (2021). An Updated Economic Analysis of the Global Polio Eradication Initiative. Risk Analysis, 41(2), 393–406. https://doi.org/10.1111/risa.13665
Ching, P., Birmingham, M., Goodman, T., Sutter, R., & Loevinsohn, B. (2000). Childhood mortality impact and costs of integrating vitamin A supplementation into immunization campaigns. American Journal of Public Health, 90(10), 1526.
Global Polio Eradication Initiative. (2016). Financial Resources Requirement. Geneva: World Health Organization. Retrieved from http://polioeradication.org/wp-content/uploads/2016/10/FRR2013-2019_April2016_EN_A4.pdf.
Retrieved from Page 24 of Global Polio Eradication Initiative. (2016). Financial Resources Requirement. Geneva: World Health Organization. Retrieved from http://polioeradication.org/wp-content/uploads/2016/10/FRR2013-2019_April2016_EN_A4.pdf.
Cite this work
Our articles and data visualizations rely on work from many different people and organizations. When citing this topic page, please also cite the underlying data sources. This topic page can be cited as:
Saloni Dattani, Fiona Spooner, Sophie Ochmann, and Max Roser (2022) - “Polio” Published online at OurWorldinData.org. Retrieved from: 'https://ourworldindata.org/polio' [Online Resource]BibTeX citation
@article{owid-polio,
author = {Saloni Dattani and Fiona Spooner and Sophie Ochmann and Max Roser},
title = {Polio},
journal = {Our World in Data},
year = {2022},
note = {https://ourworldindata.org/polio}
}Reuse this work freely
All visualizations, data, and articles produced by Our World in Data are completely open access under the Creative Commons BY license. You have the permission to use, distribute, and reproduce these in any medium, provided the source and authors are credited.
The data produced by third parties and made available by Our World in Data is subject to the license terms from the original third-party authors. We will always indicate the original source of the data in our documentation, so you should always check the license of any such third-party data before use and redistribution.
All of our charts can be embedded in any site.