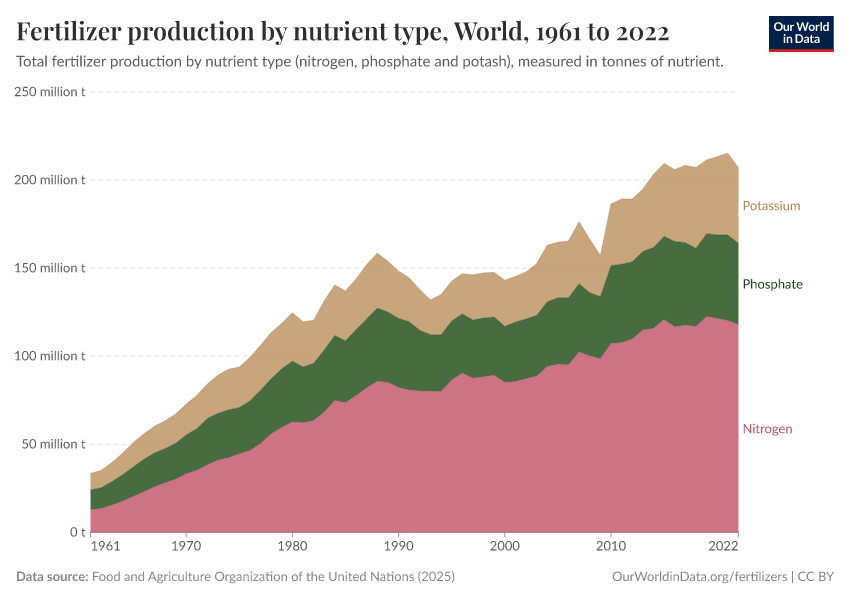
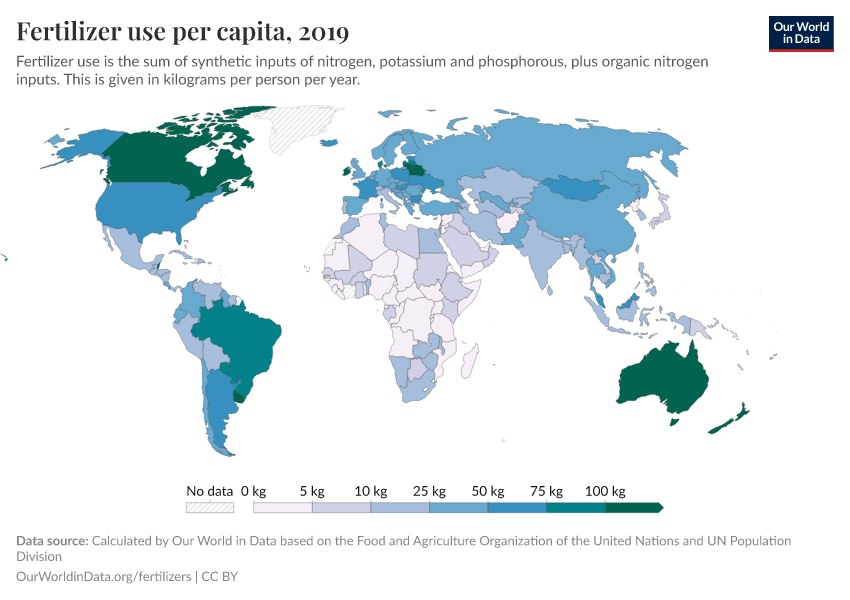
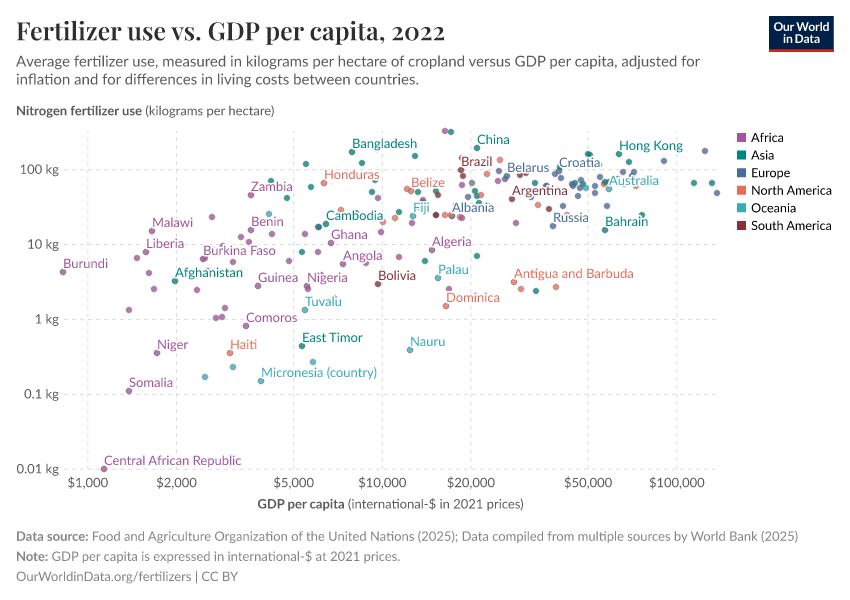
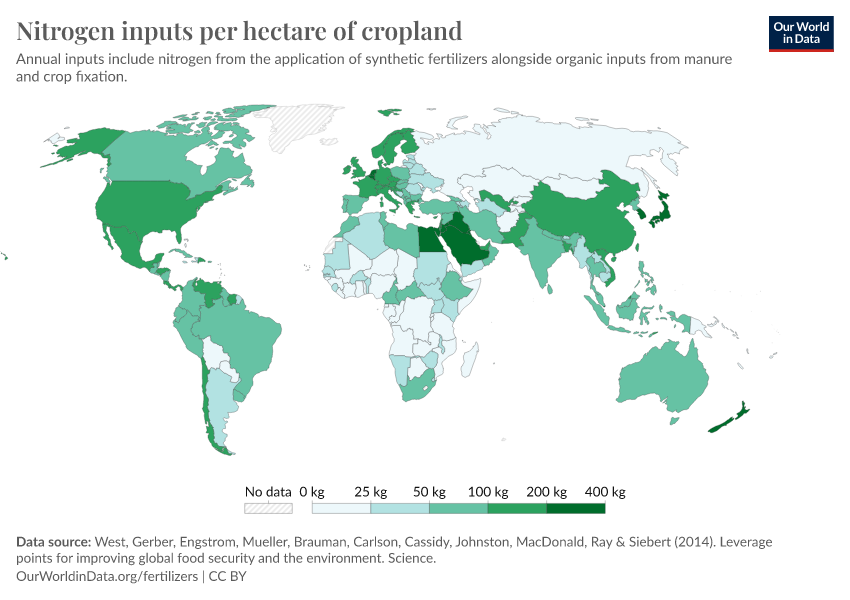
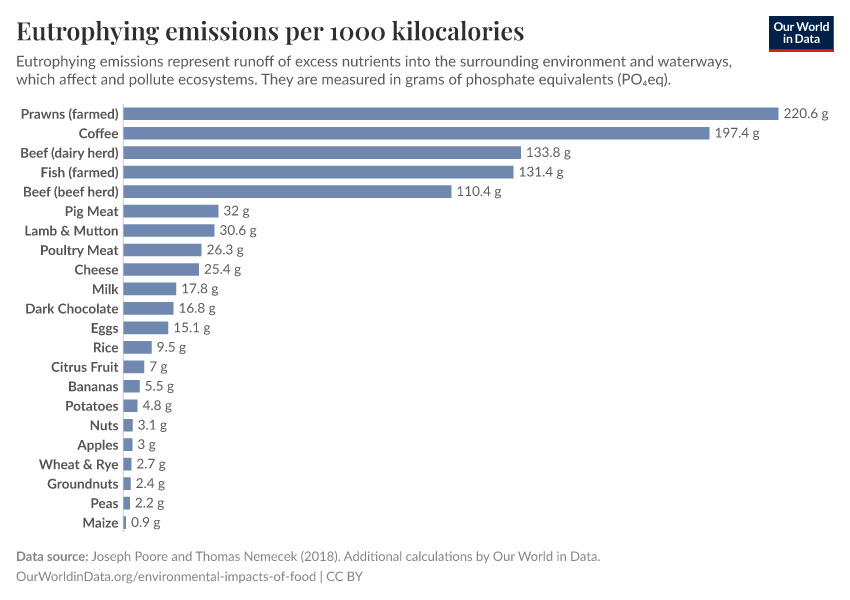
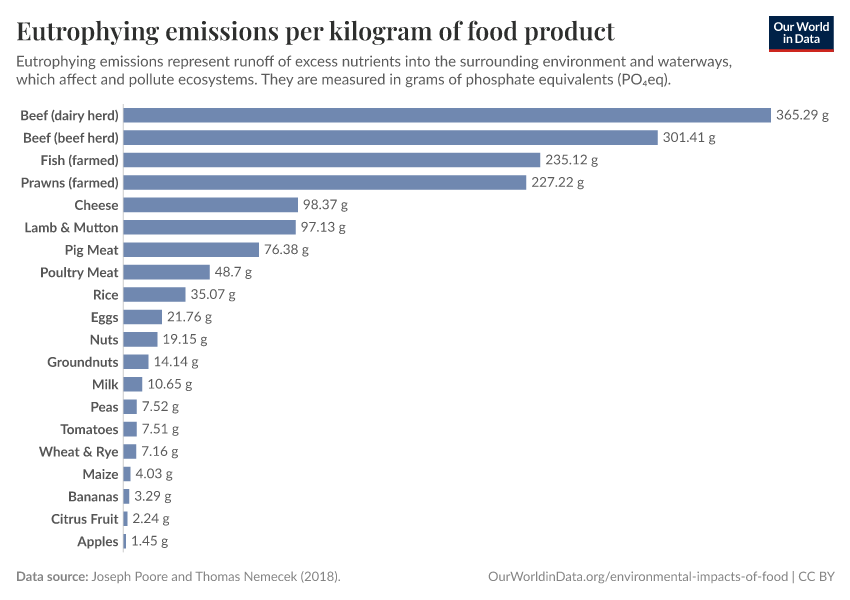
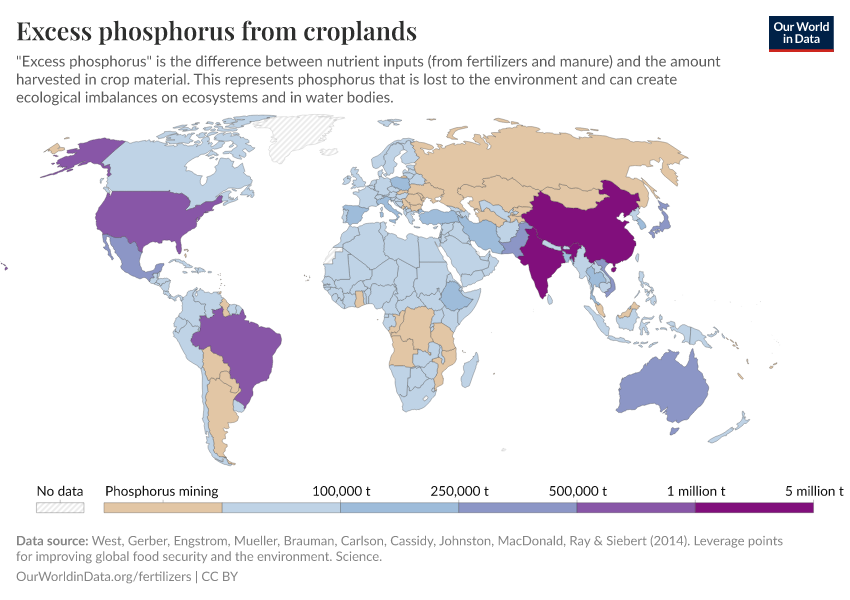
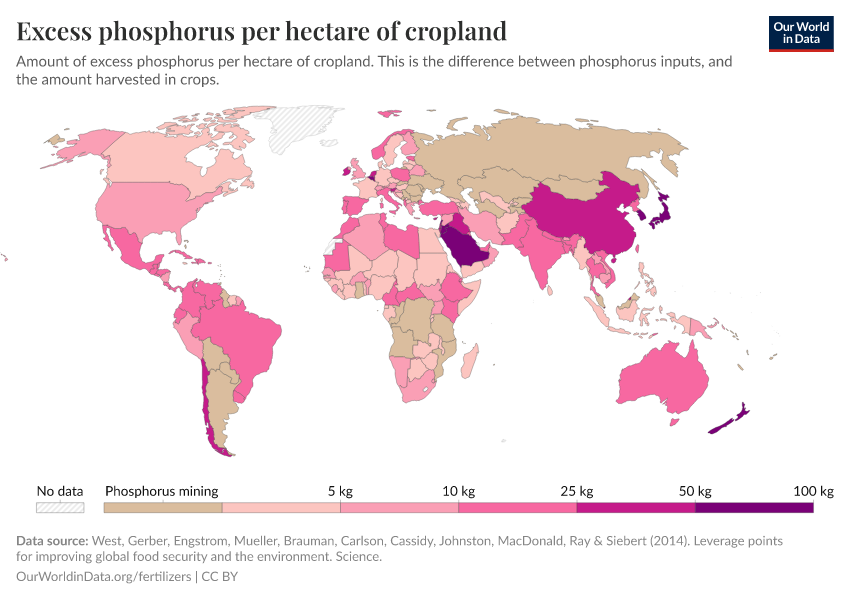
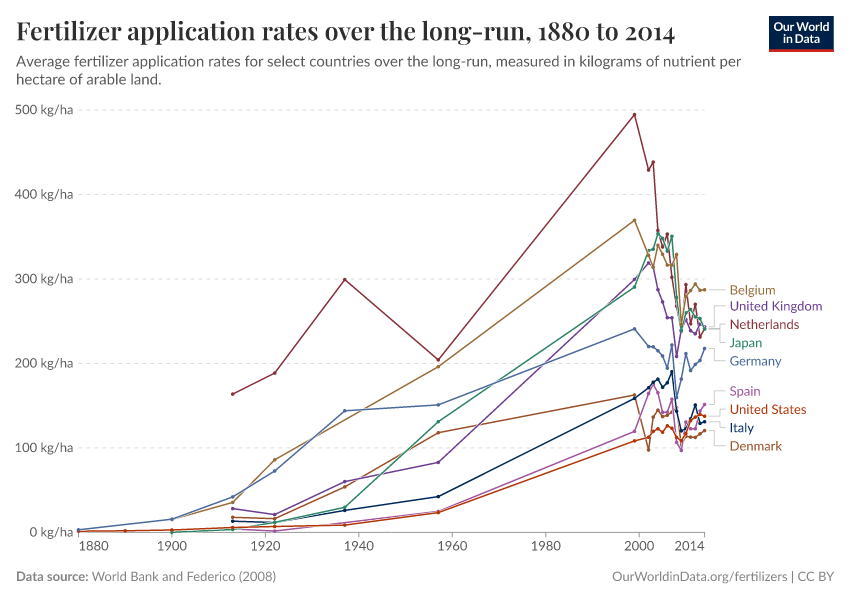
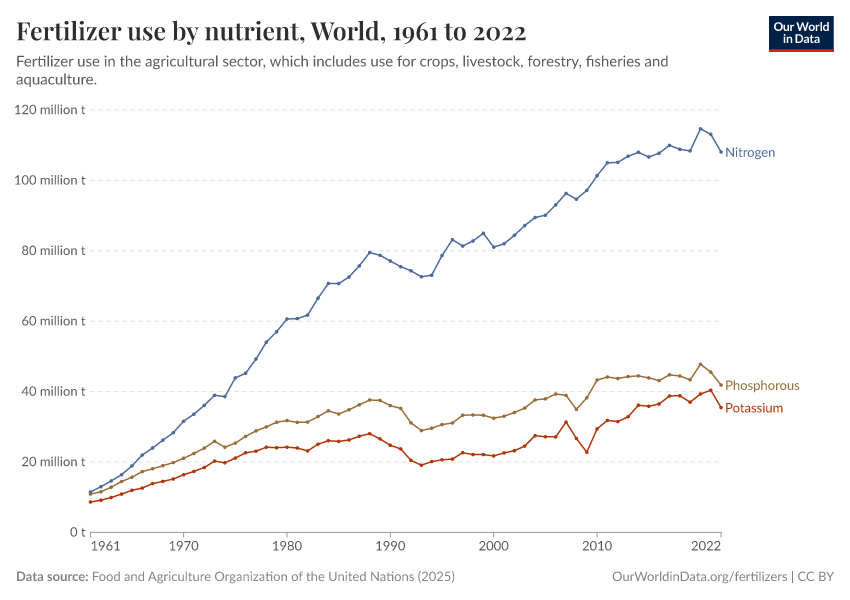
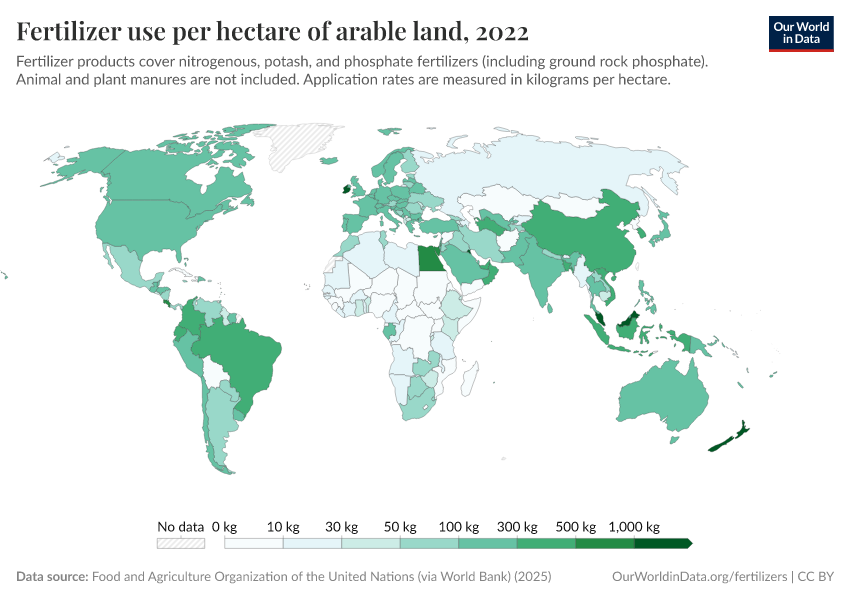
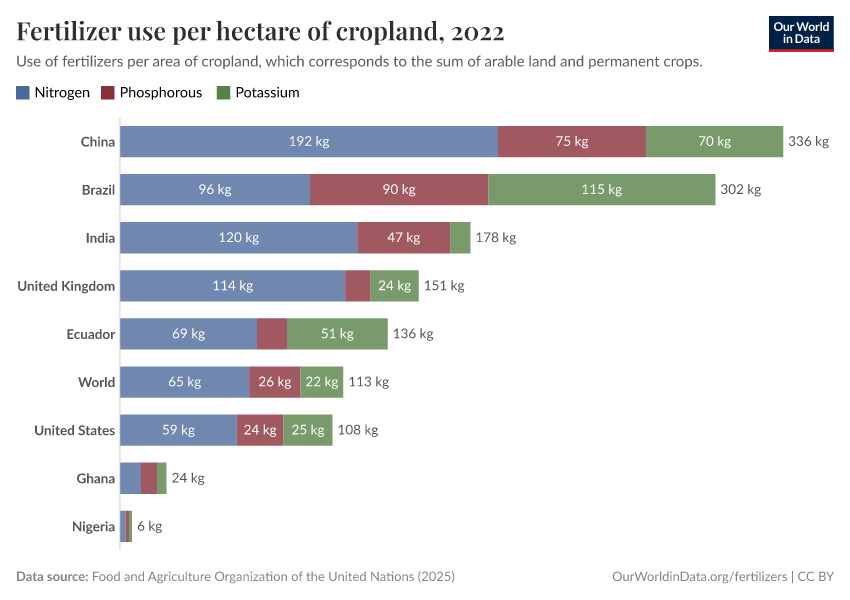
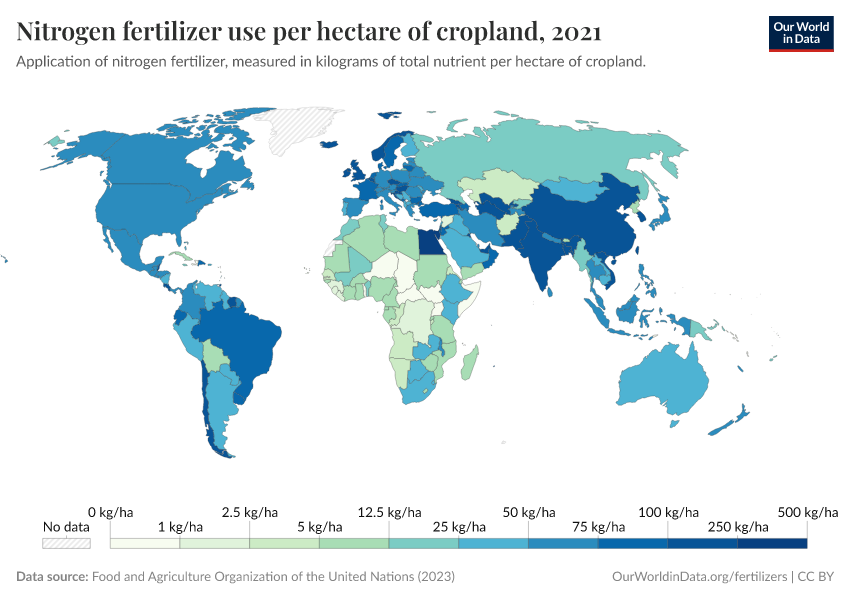
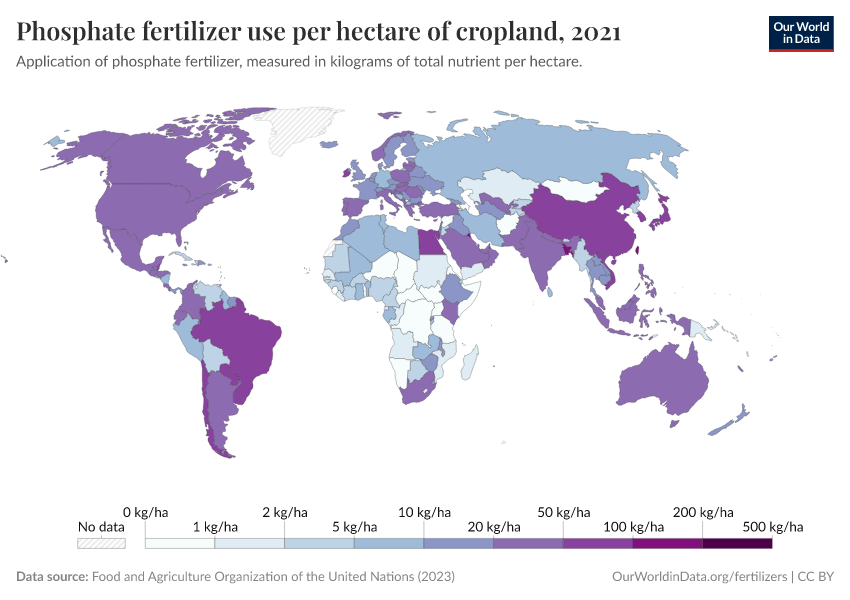
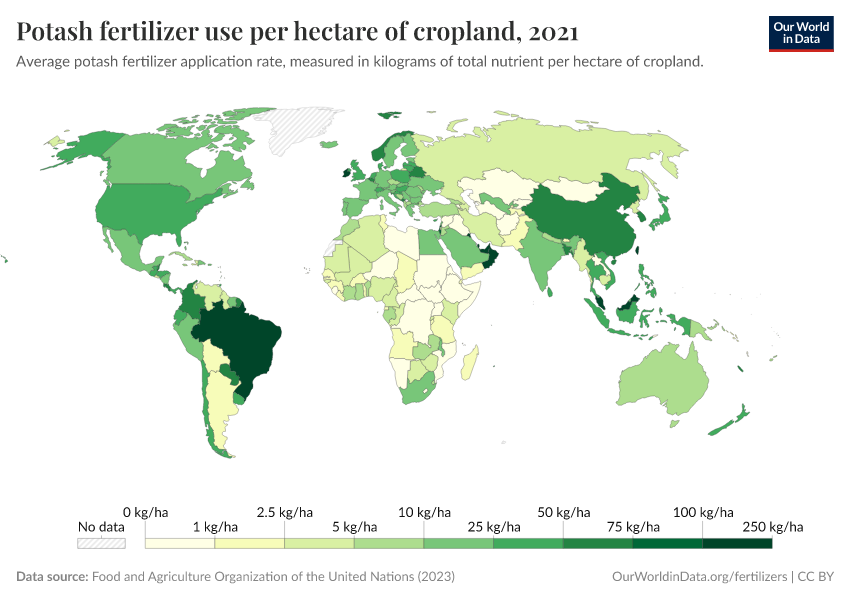
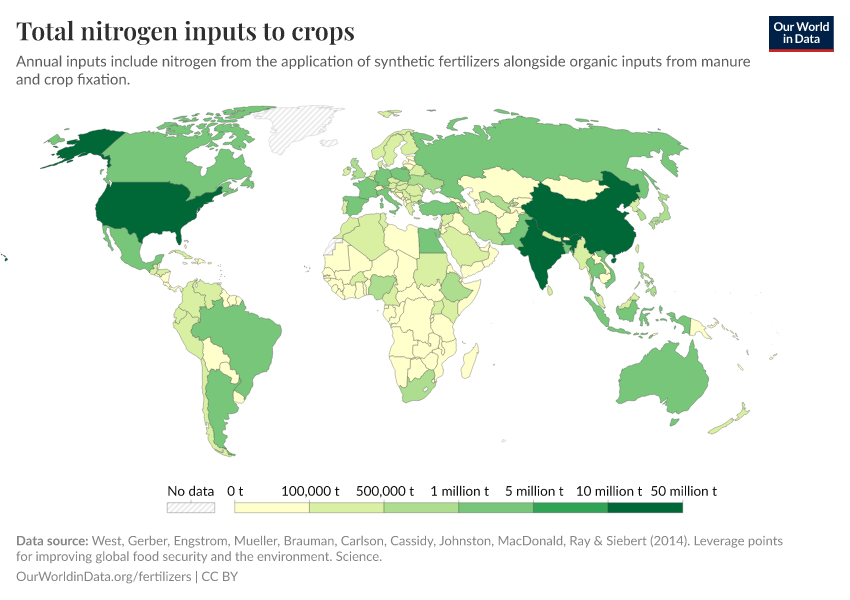
Fertilizers
Fertilizers supply plants with nutrients that are essential for growth.
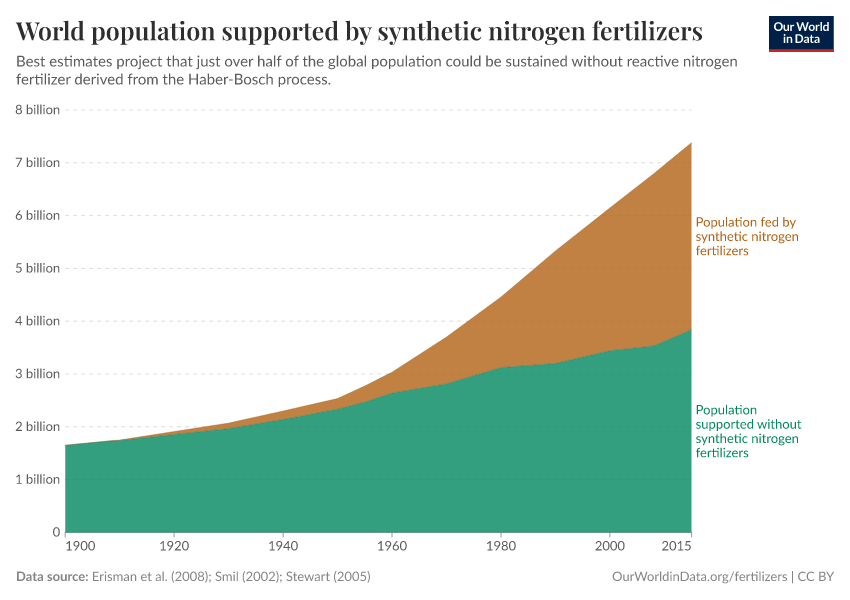
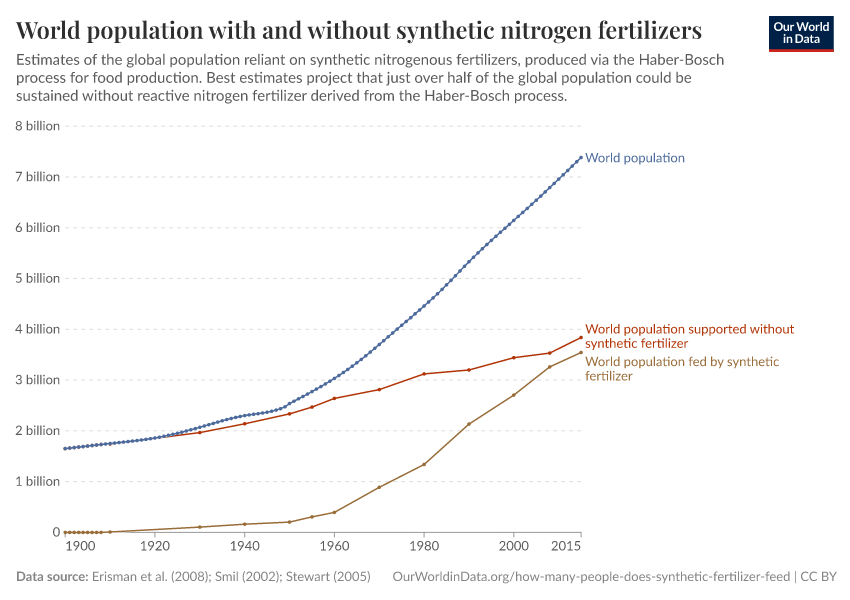
Fertilizers have played an essential role in feeding a growing global population. It's estimated that just under half of the people alive today are dependent on synthetic fertilizers.
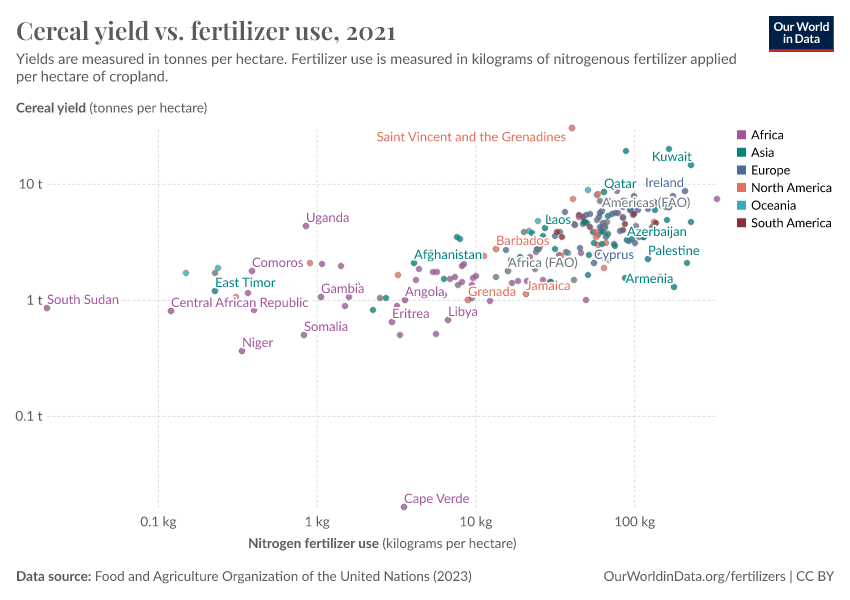
They can bring environmental benefits too: fertilizers can increase crop yields. By increasing crop yields we can reduce the amount of land we use for agriculture.
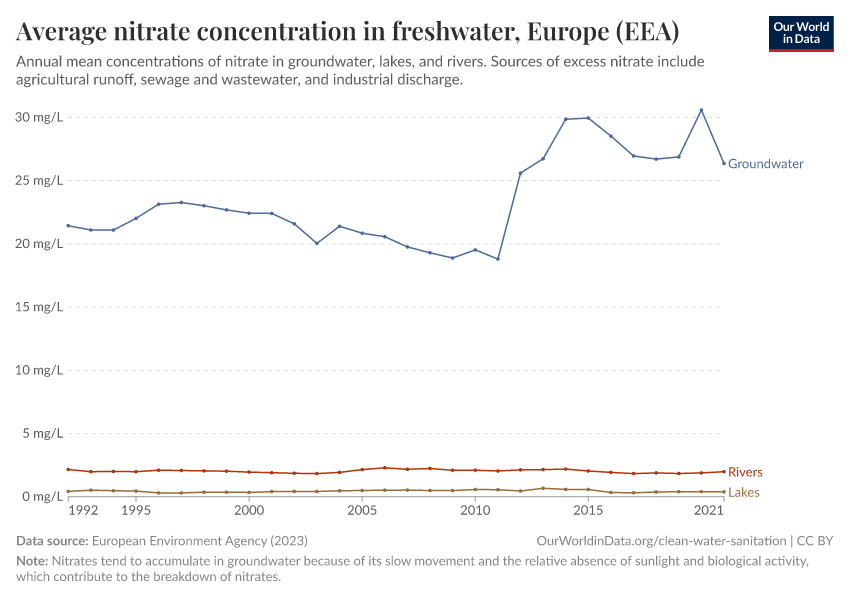
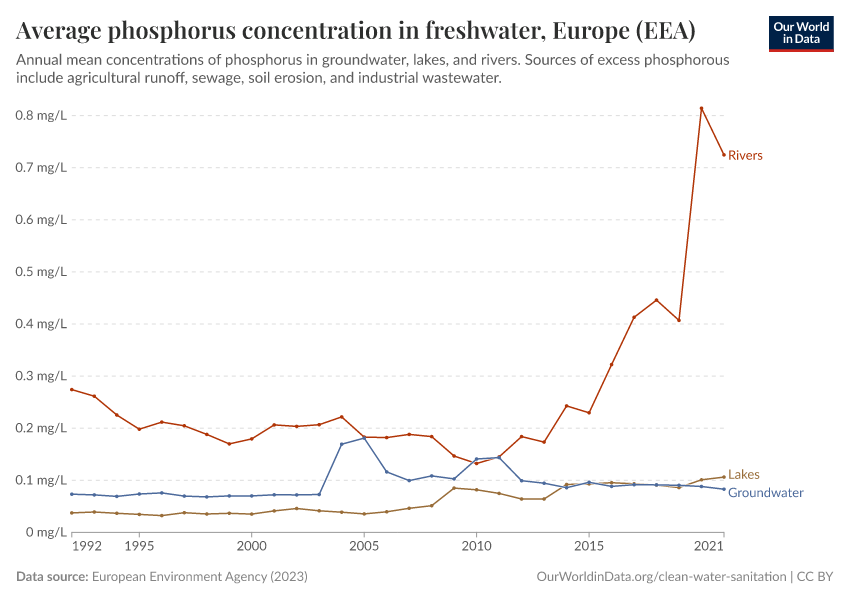
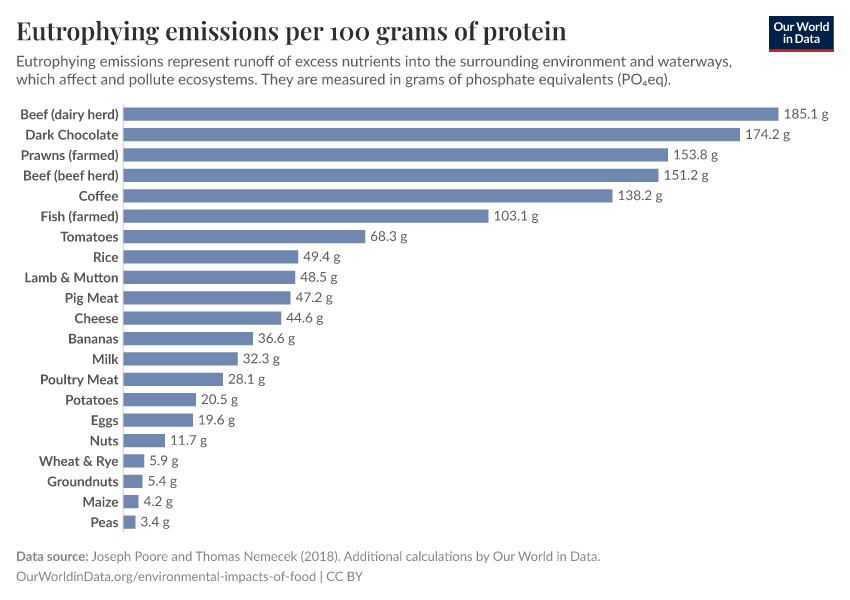
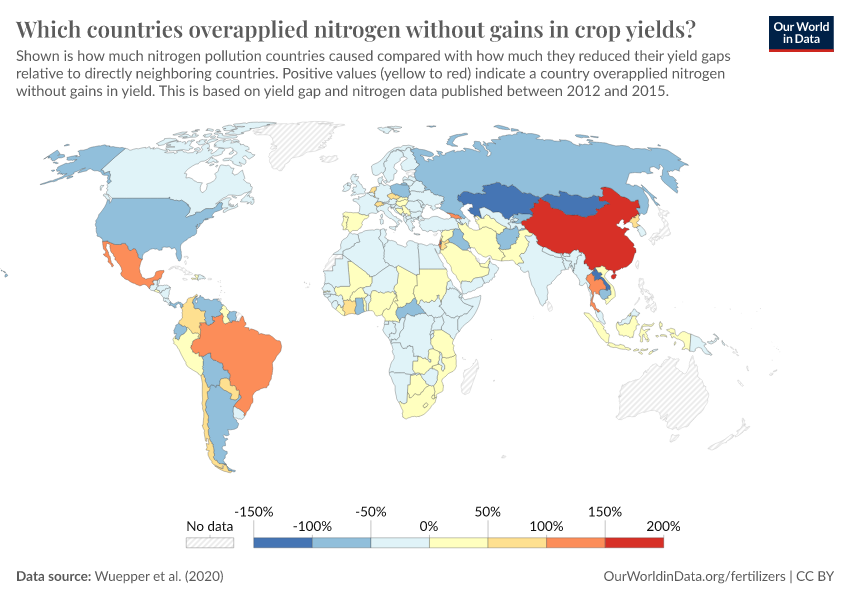
But they also create environmental pollution. Many countries overapply fertilizers, leading to the runoff of nutrients into water systems and ecosystems.
A problem we need to tackle is using fertilizers efficiently: yielding its benefits to feed a growing population while reducing the environmental damage that they cause.
On this page you can find all our data, visualizations, and writing relating to fertilizers.
Explore data on Fertilizers
Research & Writing
How many people does synthetic fertilizer feed?
It's estimated that around half of the world population today are reliant on fertilizer for food production.
Can we reduce fertilizer use without sacrificing food production?
Some countries need more fertilizers to increase crop yields. But some could cut back without sacrificing food production.
More key articles on Fertilizers
Excess fertilizer use: Which countries cause environmental damage by overapplying fertilizers?
Increasing agricultural productivity across Sub-Saharan Africa is one of the most important problems this century
August 22, 2017
Yields vs. land use: how the Green Revolution enabled us to feed a growing population
Key Charts on Fertilizers
See all charts on this topicFeatured Data on Fertilizers
Cite this work
Our articles and data visualizations rely on work from many different people and organizations. When citing this topic page, please also cite the underlying data sources. This topic page can be cited as:
Hannah Ritchie, Max Roser, and Pablo Rosado (2022) - “Fertilizers” Published online at OurWorldinData.org. Retrieved from: 'https://ourworldindata.org/fertilizers' [Online Resource]BibTeX citation
@article{owid-fertilizers,
author = {Hannah Ritchie and Max Roser and Pablo Rosado},
title = {Fertilizers},
journal = {Our World in Data},
year = {2022},
note = {https://ourworldindata.org/fertilizers}
}Reuse this work freely
All visualizations, data, and articles produced by Our World in Data are completely open access under the Creative Commons BY license. You have the permission to use, distribute, and reproduce these in any medium, provided the source and authors are credited.
The data produced by third parties and made available by Our World in Data is subject to the license terms from the original third-party authors. We will always indicate the original source of the data in our documentation, so you should always check the license of any such third-party data before use and redistribution.
All of our charts can be embedded in any site.