Smallpox
Humanity eradicated this infectious disease globally. How was this possible?
This article was first published in June 2018, and we updated the text in February 2024.
Smallpox is the only human disease that has been successfully eradicated.1
It is an infectious disease caused by the variola virus, and was a major cause of mortality in the past, with historic records of outbreaks across the world. Its historic death tolls were so large that it is often likened to the bubonic plague.
The eradication of smallpox is therefore a major success story for global health. It was a disease that was endemic across all continents and caused major mortality worldwide, and addressing it led to crucial advances in the field of immunology. The smallpox vaccine was the first vaccine to be developed.
See all interactive charts on smallpox ↓
Related topics
Other research and writing on smallpox on Our World in Data:
Smallpox: disease, transmission & symptoms
What is smallpox?
Smallpox is an infectious disease that is caused by the variola virus. It spreads from one person to another, either directly or indirectly. The variola virus infects only humans — other animals are unable to catch smallpox.
There are two types of variola viruses — variola major and variola minor — with the former type being much more severe and deadly.
How is it transmitted?
People can be infected with the variola virus if they inhale droplets, or come into contact with droplets, containing the virus from someone who is infected with it. Coughing and sneezing, for example, can lead to the virus being spread through droplets.2
Symptoms
The classic symptom of smallpox is the rash — these are pustules covering a patient's entire body. After being infected with the variola virus, patients usually have no symptoms for 10–14 days (which is called the incubation period) followed by symptoms like a fever and headache — which resemble a common cold — for 2–3 days. Since these symptoms are not specific to smallpox, it can be very difficult to tell from symptoms alone that they have been infected.3
On average, it then takes 24 hours for the rash to cover the body, and an additional three weeks for them to grow in size, form crusts, and eventually fall off. A surviving patient would be marked for life with depigmented skin and scars in places where pustules had formed.
Age profile of patients
Smallpox could infect people of any age. However, in societies where it was endemic, it mainly affected children. Adults in an endemic population would have been infected with the virus at an earlier stage in their lives already and since the only two possible outcomes of an infection were death or survival and life-long immunity, the virus could only infect and be transmitted by individuals that had never been exposed before: children.
In 17th century Britain, this led to children not being considered full members of the family until they had survived their smallpox infection.4
Death
Among people known to be infected with the variola major virus, around 30% are estimated to have died. In contrast, an infection of the variola minor virus was milder — it would lead to death in less than one percent of cases.5
The specific way a smallpox infection would lead to a patient's death remains unclear.2 The Oxford Textbook of Medicine notes that people infected may develop symptoms of prostration (total exhaustion), toxemia (toxins in the blood), and hypotension (low blood pressure), which all contribute, but the mechanism by which smallpox causes such symptoms are not clear.6
Among those who died from smallpox, death usually occurred within 10 to 16 days of the beginning of their symptoms.7
Patients who successfully fought off the infection and survived, would likely never catch smallpox again.8 They would develop long-lasting immunity to the disease, through the production of memory cells that could quickly detect and activate killer cells against variola viruses that they encountered in the future.
Lack of treatment
While smallpox was endemic, there was never a treatment for it. Once a person was infected, it wasn’t possible to treat them — one could only helplessly let the disease run its course and hope they survived.
Modern antiviral drugs might now allow treatment of the disease if the disease still existed — the Centers for Disease Control and Prevention (CDC)9 now lists three antiviral drugs as potential treatment options.
Origins of smallpox
The origin of the variola virus and the time since when it infected humans are uncertain.
Classic symptoms of smallpox — the skin eruptions and pustules — have been identified in mummified individuals dating back to 1100–1500 BCE in Egypt.10
One of these was Pharaoh Ramses V who died in 1157 BCE. The photograph of his skull below shows small pustules, especially on his cheeks.11
The origins and evolution of smallpox have been explored further using paleogenomic evidence, which involves sequencing ancient genomes from mummified individuals and other samples from burials, and analyzing them to see how closely related they are and when they likely shared a common genetic ancestor.13
The first ancient genome, extracted from a 400-year-old Lithuanian child, suggested that the major divergence between modern variola virus strains occurred within the last two centuries.14
But four other genomes, from Viking-era samples dating from 600 to 1050 CE, were later discovered, and analyses of these samples suggested that the divergence actually occurred around 1700 years ago. They also revealed an extinct lineage, which suggested that smallpox was more widely spread across Europe than historical records suggested.15
Ancient DNA has been important in our understanding of smallpox's evolutionary history, but genomic analysis in dating the virus's origins and evolution can still be challenging.
One reason is that, when vaccination first emerged it led to significant evolutionary pressure on the variola virus, and caused some of its strains to go extinct. Another reason is that we may not have uncovered a wide enough diversity of ancient samples yet. These factors can skew genetic estimates, and complicate efforts to understand when the virus actually emerged.16 They also highlight the need for further research to better understand the origins and history of the variola virus.
Empirical View
How many died of smallpox?
Donald Henderson — who directed the WHO’s program to eradicate smallpox worldwide — reports that, during the 20th century alone, "an estimated 300 million people died of the disease" in his review paper “The eradication of smallpox — An overview of the past, present, and future”.17
He also suggests that in the last hundred years of its existence smallpox killed “at least half a billion people.”18 Over a century, this translates to around 5 million annual deaths on average.
Long-run smallpox deaths in Europe
The graph shows the death rate over the long run in some European countries. The data spans the period from 1774 to 1900 in European countries for which data was available.
Two phenomena are illustrated here.
First, Swedish data, which pre-dates the smallpox vaccine in 1796, shows the large disease burden and endemicity of smallpox in the 18th century. In peak years, up to 7 out of 1000 people in Sweden died of smallpox, and the endemic disease had volatile outbreaks.
Second, countries that had adopted vaccination legislation earlier saw their smallpox death rates decline more substantially and suffered fewer casualties during the Franco-Prussian War of 1870–1871.
Austria and Belgium — neither of which ever made smallpox vaccination mandatory — still recorded large numbers of smallpox deaths from 1875 onwards, while other European countries had already driven down smallpox fatalities significantly by then.
Even so, smallpox remained endemic in every European country at the end of the 19th century.

Number of smallpox cases
Smallpox cases by country
The chart depicts the reported number of smallpox cases by most countries between 1920 and 1977.
You can see that, by the time the World Health Organization (WHO) launched its Intensified Smallpox Eradication Program in 1966, most countries in Western Europe and North America had almost eliminated smallpox.
However, countries across South America, Africa, and Asia, especially India, still recorded very large numbers — note the large differences in the color brackets below.

Global decline of smallpox
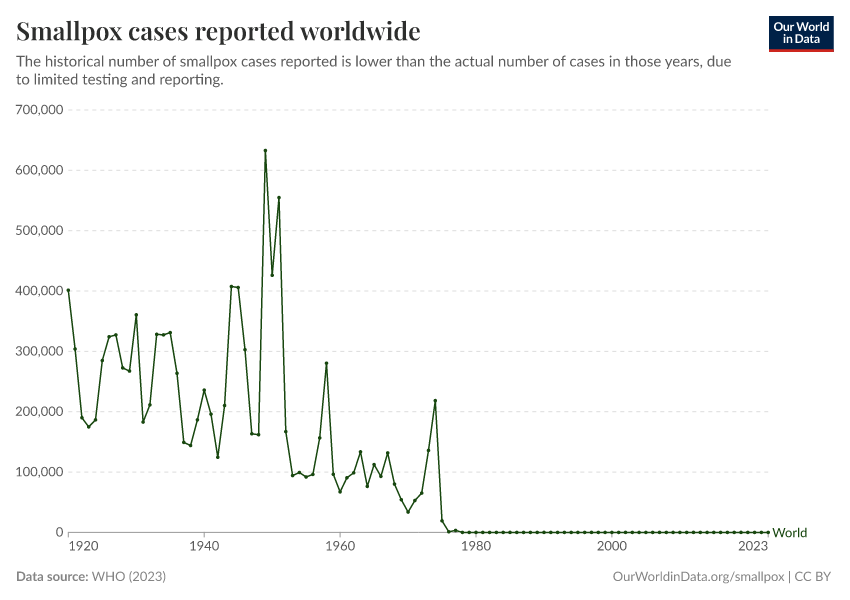
Global data on the number of smallpox cases is shown in the chart. Shown here is the number of reported smallpox cases worldwide from 1920 until the last case in 1977.
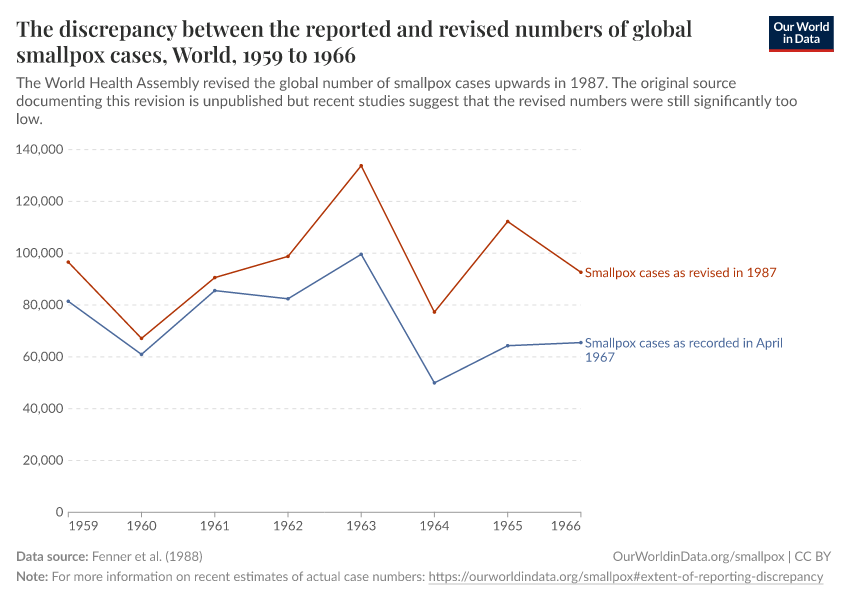
The reported number of smallpox cases between 1920 and 1978 amounted to 11.6 million cases — but that number was certainly smaller than the actual number of cases, although we do not know by how much.
Even though smallpox was easily visible and should therefore be relatively easy to document, there was not an international organization dedicated to collating this data before then, which means the number of reported cases could be substantially lower than the actual number of cases.
Crosby (1993) estimates that in 1967, 10–15 million people were still being infected with smallpox every year. In contrast, the chart on the reported cases below indicates only 132,000 for that same year.19
Read more in our article:
Smallpox decline by region
The chart below shows the number of reported cases, broken down by world region, until 1977, the year of the last case of smallpox.
Even though absolute numbers are almost certainly far too low (as explained above), it can be seen that South Asia had by far the highest number of smallpox cases.
This was primarily driven by incidence in India: in 1973, for instance, in a state that had reported approximately 500 cases a week, the WHO search team found that there were actually 10,000 cases.20

Eradication of smallpox
The last variola major infection was recorded in Bangladesh in October 1975, while the last variola minor infection occurred two years later in Merka, Somalia, on October 26th, 1977.
During the following two years, WHO teams searched the African continent for further smallpox cases among people with rash-like symptoms, which can be a symptom of several diseases. They found no further cases of smallpox.
In 1978, a laboratory accident in the United Kingdom occurred where a variola virus sample was spread through the university laboratory's ventilation system, infecting two people and killing one.21
The world map shows the year in which each country recorded the last endemic case of smallpox. As you can see, Europe, North America and Australia managed to eliminate smallpox relatively early, most by the 1940s. This predated the WHO's Intensified Smallpox Eradication Program, which was launched in 1966.
Countries across Sub-Saharan Africa, Latin America and Asia eliminated smallpox several decades later, in the 1960s and 1970s.
In May 1980, the World Health Assembly, the governing body of the World Health Organization, officially certified the global elimination of smallpox, the first-ever eradication of a disease in human history.
It further "recommended that all countries cease vaccination, all laboratories should destroy their remaining stocks or transfer them" to one of two certified high-security laboratories. One was in the Vector Institute in Siberia and another was the Centers for Disease Control and Prevention (CDC) in Atlanta.22
These are the only two known places that still hold samples of the variola viruses for research purposes.

How was global decline & eradication achieved?
Variolation
Discovery of variolation
Progress against smallpox began with variolation, which refers to deliberately infecting people with weakened or milder versions of the smallpox virus. It is sometimes also inoculation.
Before the year 1000, people in India and China had already observed that children who survived smallpox infections were protected against any future outbreaks of the disease. As a consequence, they developed a procedure that involved the nasal inhalation of dried smallpox scabs by three-year-olds.2
Another commonly practiced technique, whose origins are unknown, involved injecting the liquid from pustules of a smallpox patient into a healthy person, underneath their skin. This would usually result in a milder infection of smallpox, and the person would then develop immunity against the disease.
Both practices became known as variolation (inoculation) techniques.
However, there were disadvantages of variolation. One was that, during the course of the mild infection, the person became a carrier of the disease and could infect other people. Additionally, it was difficult to control the severity of the infection — it could sometimes develop into a full-blown smallpox case that could lead to death.23
This meant that the practice usually reduced the severity of an infection and the likelihood of deaths, but was risky and couldn’t eliminate the virus.
Institutionalized variolation
A British ambassador's wife, Lady Mary Wortley Montague (1689–1762) was the force that pushed for government-mandated variolation in England.
She had suffered a smallpox infection when she was younger and had lost her younger brother to the disease at the age of 26. She first learned about variolation when she arrived in 1717 in Istanbul, where variolation was commonly practiced. She later had the embassy inoculate her two children.
News about the practice spread among the royal family. After following trials, Charles Maitland, a physician, successfully inoculated the two daughters of the Princess of Wales with variolation in 1722. Thereafter, variolation became a common practice in Great Britain and became known in other European countries.
It became an even more established practice when the French King Louis XV died of smallpox in May of 1776 and his successor and grandson Louis XVI was inoculated with the variola virus one month later.
Vaccine against smallpox
At the end of the 18th century, British surgeon and physician Edward Jenner (1749-1823) pioneered the first-ever vaccination against an infectious disease.
He had been inoculated through variolation at the age of 8, and later as a surgeon, variolation was part of his work.24
He also observed that people who had suffered from cowpox would subsequently have a very mild, if at all visible, reaction to the smallpox variolation. This was because the cowpox and variola viruses were members of the same virus family, called orthopox viruses, and provided immunity against each other, which he had unknowingly observed.
He hypothesized that variolation using the cowpox virus would protect children against smallpox as well. Since cowpox infections were much milder and never fatal, this would eliminate the problem of variolated children being carriers of smallpox and sometimes dying of the virus developing into a full-blown infection. In addition, it could reduce the pool of humans that the variola virus needed to survive, and it brought elimination, and eventually eradication, of smallpox into the realm of possibility.
In May 1796, Jenner inoculated a boy with cowpox, and then a few months later, with the smallpox virus. When the boy did not develop any smallpox symptoms in response to being variolated, his hypothesis that cowpox offered protection from smallpox was confirmed, motivating his further research trials.
Initially, Jenner faced major barriers to spreading the word about his discovery. When he submitted a paper outlining his findings to the journal Philosophical Transactions edited by the Royal Society, it was rejected. They even advised him not to pursue his ideas any further, pointing to the detrimental impact on his career and reputation. Undeterred, he published his work with an increased number of trials at his own expense two years later, in 1798. He also went on to convince colleagues and supply them with vaccines in other British cities of his new procedure that became known as vaccination (derived from the Latin word for cow, Vacca).
By 1802, the British Parliament did acknowledge his important contribution and awarded him £30,000. Meanwhile, vaccination had spread to most of Europe and New England.25
His 1798 publication Inquiry into the Variolae vaccinae known as the Cow Pox had been translated into German, French, Spanish, Dutch, Italian, and Latin within three years. US President Thomas Jefferson was an important figure in the widespread application of vaccination throughout the United States, and in 1806, he thanked Edward Jenner in a letter for his discovery and famously predicted "Future generations will know by history only that the loathsome smallpox existed and by you has been extirpated."26
The dramatic decline in smallpox fatalities in response to Jenner's vaccine can be traced in the chart, which shows the number of deaths due to smallpox as a share of all deaths in London from 1629 to 1902. Before the introduction of a smallpox vaccine in 1796, around 7% of all deaths annually were caused by smallpox. Following the introduction of the vaccine, a clear decline in smallpox deaths is visible.

Smallpox Eradication Program
It was only with the establishment of the World Health Organization (WHO) that international quality standards for the production of smallpox vaccines were introduced. This shifted the fight against smallpox from a national to an international agenda. It was also the first time that global data collection on the prevalence of smallpox was undertaken.
By 1959, the World Health Assembly, the governing body of the World Health Organization (WHO), had passed a resolution to eradicate smallpox globally. But it was not until 1966, that funding was provided to the "Intensified Smallpox Eradication Program" to increase efforts for smallpox eradication.
By 1966, the number of infections of smallpox had already substantially been reduced by national governments' efforts. Nonetheless, skepticism about the feasibility of eradication prevailed, as some previous efforts to eradicate diseases — such as malaria and yaws — had failed. Furthermore, funding for the Intensified Smallpox Eradication Programme was insufficient to meet global needs, which resulted in vaccine shortages.27
In addition, continued globalization and the growth of international air travel resulted in the continual re-introduction of the disease into countries that had previously managed to eliminate smallpox.
Overcoming the last mile problem: ring vaccination
Smallpox's eradication was greatly spurred by making use of the fact that smallpox transmission occurs via air droplets.
Initially, the WHO had pursued a strategy of mass vaccination which attempted to vaccinate as many people as possible, hoping that herd immunity (explained in our vaccine topic page) would protect the whole population. Soon, however, vaccination efforts were targeted locally around smallpox cases, because smallpox was transmitted by sick patients' air droplets. This practice is known as “ring vaccination”.
People who had been in direct contact with a smallpox patient over the last two weeks were quarantined and vaccinated. The downside of such an approach was that the virus could spread easily if it was re-introduced from overseas. This was the case in Bangladesh, for example, which had previously eliminated smallpox until 1972 when it was brought back from across its border with India.28
Despite the risk of re-introductions, ring vaccination greatly reduced the cost of the eradication campaign. The number of administered vaccines declined and smallpox was increasingly brought under control.2
One of the last strongholds of the variola virus was India. While 58% of global reported smallpox cases were reported in India in 1973, this increased to 86% in 1974.29 Major pushes in vaccination campaigns, however, successfully drove down the number of infections to zero in India in 1976.
Correlates & consequences
Impact on life expectancy
Did smallpox variolation and vaccination against smallpox have a notable impact on life expectancy?
Many have claimed that inoculation against smallpox was one of the first measures that had a positive effect on life expectancy. Angus Deaton (2013) makes this claim based on a book by Razzell (1977), which reviews existing birth, baptism and burial records in various counties of 18th century Britain.30 31
It is hard to prove such claims because of the limited amount of high-quality data on both life expectancy and smallpox cases or deaths before the smallpox vaccine was developed in 1796 and adopted across countries.
The high case fatality rate of approximately 30% (of the variola major virus) meant that smallpox shortened the lives of many. Since the virus predominantly affected children, smallpox deaths are likely to have had a disproportionate impact on average life expectancy.
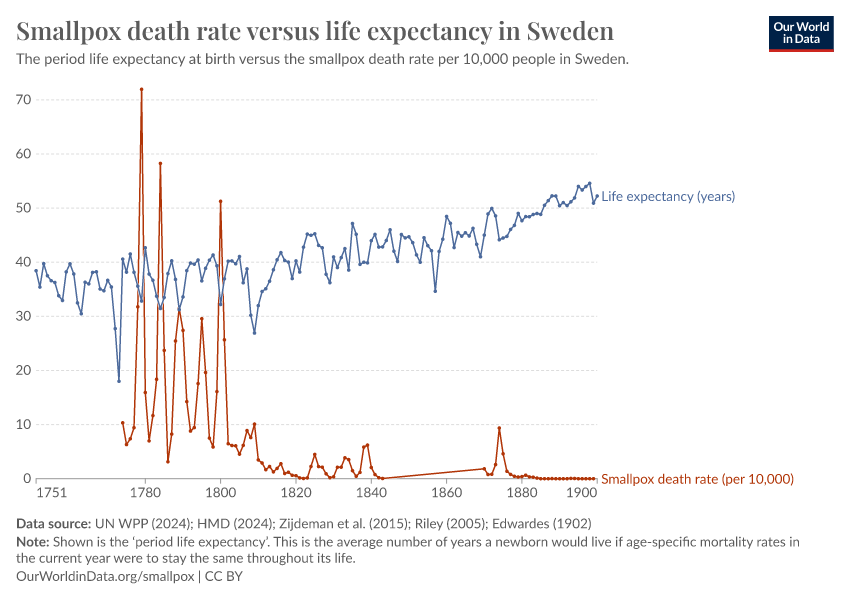
The chart below shows Sweden's life expectancy versus deaths from smallpox from 1774 to 1900.
Large smallpox outbreaks were documented in the years 1779, 1784, 1789, 1795 and 1800. These years coincide with sharp declines in life expectancy, implying that smallpox endemicity did have a substantial effect on life expectancy.
Furthermore, the available data show dramatic declines in mortality in London as well as in Sweden (below) from 1800 onwards, after the vaccine was developed.
Jenner's vaccine appears to have had a notable impact on increased life expectancy but this, to our knowledge, has not been studied in significant detail.32
Costs of smallpox and its eradication
By the time the World Health Organization launched the Intensified Smallpox Eradication Program in 1967 many countries, most of them high income countries, had already eliminated smallpox. Therefore, the total cost of eradicating smallpox is very challenging to estimate — as individual country programs reach back to before records of public health expenditure existed.
Nonetheless, the Intensified Smallpox Eradication Program has been estimated to have cost $300 million in total from 1967 to 197733 with one-third of the funding provided by international donors and the remaining two-thirds financed by endemic country governments.34
Beyond direct program costs, smallpox incurred much higher indirect costs in the form of foregone economic performance.
It has been estimated that smallpox cost low-to-middle income countries more than $1 billion per year at the beginning of the Intensified Smallpox Eradication Program in 1967, with more than $20 million dedicated to the care of infected patients.35 Industrialized countries incurred much lower costs: $350 million in 1968, which included vaccination programs and absence from work costs.36
Overall, the Center for Global Development37 estimated that direct and indirect costs of smallpox cost the world approximately $1.35 billion in the late 1960s.
Following its eradication, countries now do not have to spend money on vaccine development and administration. Estimates on the savings from forgone costs thanks to the eradication of smallpox exist, but we are not aware of estimates that we would consider reliable.38
Key Charts on Smallpox
See all charts on this topicFeatured Data on Smallpox
Endnotes
While one other disease, Rinderpest, has also been eradicated, smallpox is the only one that infected humans. Rinderpest "only" infected animals, predominantly cattle and buffalo, and was declared eradicated in 2011.
Atkinson, W., Hamborsky, J., McIntyre, L., & Wolfe, C. (2007). Appendix Chapter on Smallpox - Epidemiology and Prevention of Vaccine-Preventable Diseases (10th ed., pp. 281-306). Atlanta: Centers for Disease Control and Prevention (CDC). Fully available online at http://www.docsimmunize.org/immunize/cdcmanual/original/smallpox.pdf.
These unspecific symptoms usually entailed a fever and headache but could also involve nausea, backaches or delirium. Page 5 of Fenner, F., Henderson, D., Arita, I., Jezek, Z., & Ladnyi, I. (1988). Smallpox and its eradication. Geneva: World Health Organization. Fully available for download here.
Kotar, S., & Gessler, J. (2013). Smallpox: A History. Jefferson, North Carolina: McFarland & Company. Partly available on google books.
30% is the fatality rate reported in Kotar, S., & Gessler, J. (2013). Smallpox: A History. Jefferson, North Carolina: McFarland & Company. Partly available on Google Books. Koplan and Foster (1979) reported a range of 15 to 45%. Koplan, J., & Foster, S. (1979). Smallpox: Clinical Types, Causes of Death, and Treatment. Journal Of Infectious Diseases, 140(3), 440-441.
Smith, G.L. (2010) Poxviruses. In D. Warrell, T. Cox & J. Firth, Oxford Textbook of Medicine (4th ed.). Oxford: Oxford University Press. Partially available online on Google Books.
Page 22 of Fenner, F., Henderson, D., Arita, I., Jezek, Z., & Ladnyi, I. (1988). Smallpox and its eradication. Geneva: World Health Organization. Fully available for download here.
People who were immunocompromised may have still been vulnerable to a repeated infection by smallpox later on.
CDC. (2018). Prevention and Treatment - Smallpox. Centers for Disease Control and Prevention. Retrieved 28 March 2018, from https://www.cdc.gov/smallpox/prevention-treatment/index.html.
Li, Y., Carroll, D. S., Gardner, S. N., Walsh, M. C., Vitalis, E. A., & Damon, I. K. (2007). On the origin of smallpox: correlating variola phylogenics with historical smallpox records. Proceedings of the National Academy of Sciences, 104(40), 15787-15792.
Page 210 of Fenner, F., Henderson, D., Arita, I., Jezek, Z., & Ladnyi, I. (1988). Smallpox and its eradication. Geneva: World Health Organization. Fully available for download here.
This picture is available on Wikimedia Commons.
Modern strains of the variola virus fall under two “clades” (phylogenetic groups). These groups are partly a result of historical events and human migrations — such as the Viking migrations, colonization, maritime exploration, the transatlantic slave trade, and the “Columbian Exchange”, which had a devastating role in population collapses in Native Americans, who were previously unexposed to smallpox.
Guzmán-Solís, A. A., Navarro, M. A., Ávila-Arcos, M. C., & Blanco-Melo, D. (2023). A Glimpse into the Past: What Ancient Viral Genomes Reveal About Human History. Annual Review of Virology, 10(1), 49–75. https://doi.org/10.1146/annurev-virology-111821-123859
Duggan, A. T., Perdomo, M. F., Piombino-Mascali, D., Marciniak, S., Poinar, D., Emery, M. V., ... & Poinar, H. N. (2016). 17th century variola virus reveals the recent history of smallpox. Current Biology, 26(24), 3407-3412.
Mühlemann, B., Vinner, L., Margaryan, A., Wilhelmson, H., de la Fuente Castro, C., Allentoft, M. E., ... & Sikora, M. (2020). Diverse variola virus (smallpox) strains were widespread in northern Europe in the Viking Age. Science, 369(6502), eaaw8977.
Duchêne, S., Ho, S. Y. W., Carmichael, A. G., Holmes, E. C., & Poinar, H. (2020). The Recovery, Interpretation and Use of Ancient Pathogen Genomes. Current Biology, 30(19), R1215–R1231. https://doi.org/10.1016/j.cub.2020.08.081
Guzmán-Solís, A. A., Navarro, M. A., Ávila-Arcos, M. C., & Blanco-Melo, D. (2023). A Glimpse into the Past: What Ancient Viral Genomes Reveal About Human History. Annual Review of Virology, 10(1), 49–75. https://doi.org/10.1146/annurev-virology-111821-123859
Henderson, D. A. (2011). The eradication of smallpox — An overview of the past, present, and future. Vaccine, 29, D7–D9. https://doi.org/10.1016/j.vaccine.2011.06.080
D. A. Henderson (2009) — Smallpox: The Death of a Disease - The Inside Story of Eradicating a Worldwide Killer. Published by Prometheus Books.
Crosby, A.W. (1993). Smallpox. In K.F. Kiple, The Cambridge World History of Human Disease (pp. 1008-1014). Cambridge: Cambridge University Press.
World Health Organization (2008) Smallpox: dispelling the myths. An interview with Donald Henderson. Bulletin of the World Health Organization 86(12). 909-988. Fully available online on the WHO website.
Lockley, M. (2016) The smallpox death that locked down Birmingham could have been avoided. Birmingham Mail. Retrieved 19 July 2018 from here.
Muula, A. S. (2022). Does the ‘world’ still need to keep live samples of the smallpox virus? Malawi Medical Journal: The Journal of Medical Association of Malawi, 34(2), 72. https://doi.org/10.4314/mmj.v34i2.1
Atkinson, W., Hamborsky, J., McIntyre, L., & Wolfe, C. (2007). Appendix Chapter on Smallpox - Epidemiology and Prevention of Vaccine-Preventable Diseases (10th ed., pp. 281-306). Atlanta: Centers for Disease Control and Prevention (CDC). Fully available online at http://www.docsimmunize.org/immunize/cdcmanual/original/smallpox.pdf
Kotar, S., & Gessler, J. (2013). Smallpox: A History. Jefferson, North Carolina: McFarland & Company Inc., Publishers. The first 92 pages are available on Google Books.
Bardell, D. (1977). Edward Jenner and the First Vaccination. The American Biology Teacher, 39(7), 440-441. The first page can be accessed here.
Riedel, S. (2005). Edward Jenner and the History of Smallpox and Vaccination. Baylor University Medical Center Proceedings, 18(1), 21-25. Fully available online on the NCBI website.
Page 248 in Magner, L. (1992). A History of Medicine. New York: Marcel Dekker. Available on Google Books.
Page 423 of Fenner, F., Henderson, D., Arita, I., Jezek, Z., & Ladnyi, I. (1988). Smallpox and its eradication. Geneva: World Health Organization. Fully available for download here.
Footnote 10 on page 372 of Kotar, S., & Gessler, J. (2013). Smallpox: A History. Jefferson, North Carolina: McFarland & Company Inc., Publishers. The first 92 pages are available on Google Books.
Hopkins, J. (1989). The Eradication of Smallpox: Organizational Learning and Innovation in International Health. Avalon Publishing.
Razzell, P. (1977). The Conquest of Smallpox: The Impact of Inoculation on Smallpox Mortality in Eighteenth Century Britain. Firle: Caliban Books.
Deaton, A. 2013. “What does the empirical evidence tell us about the injustice of health inequalities?.” Inequalities in health: concepts, measures and ethics (Nir Eyal, Samia Hurst, Ole Frithof Norheim, and Daniel Wikler, editors). Oxford, UK : Oxford University Press. Freely available online on Angus Deaton's website. Deaton, A. (2013). The Great Escape: health, wealth and the origins of inequality. New Jersey: Princeton University Press. Partly available on google books.
In addition to the mentioned studies, Riley (2001) was also reviewed.
Riley, J. (2001). Rising life expectancy: A Global History. Cambridge (UK): Cambridge University Press. Partly available on google books.
Ehreth, J. (2003). The value of vaccination: a global perspective. Vaccine, 21(27-30), 4105-4117.
Center for Global Development (undated) Case Study 1: Eradicating Smallpox. Available online on their website.
The calculations are based on the cost of caring for a smallpox patient ($2.85 in India), a person's economic productivity over their life time and each developing countries' population as well as the estimated number of smallpox cases and deaths. The number of estimated number of deaths because of smallpox used was 1.5 million. Note that this differs from the reported number of smallpox cases in 1967 which was as low as 122,000. See our section on Data Quality for discussion of this discrepancy. Calculations by Center for Global Development (undated) Case Study 1: Eradicating Smallpox. Available online on their website.
Vaccinating one person in the US against smallpox was estimated to cost $6.50; in 1968 the US spent US-$92.8 million on primary vaccinations and revaccinations against smallpox alone. Being absent from work was estimated to cost $0.75 per person per day. Calculations by Center for Global Development (undated) Case Study 1: Eradicating Smallpox. Available online on their website.
Center for Global Development (undated) Case Study 1: Eradicating Smallpox. Available online on their website.
These estimates take into account the benefits of avoided smallpox deaths and vaccination costs among other things.
Ehreth (2003) estimates that the savings from forgone smallpox deaths and vaccination costs amount to $2 billion per year globally. However it is not clear to us how the author arrived at these estimates.
CGD (undated) suggest that the US is saving its contributions to the smallpox campaign every 26 days. We were again not able to find the calculation that would back up this claim.
Ehreth, J. (2003). The value of vaccination: a global perspective. Vaccine, 21(27-30), 4105-4117.
Center for Global Development (undated) Case Study 1: Eradicating Smallpox. Available online on their website.
Cite this work
Our articles and data visualizations rely on work from many different people and organizations. When citing this topic page, please also cite the underlying data sources. This topic page can be cited as:
Sophie Ochmann, Saloni Dattani, and Max Roser (2018) - “Smallpox” Published online at OurWorldinData.org. Retrieved from: 'https://ourworldindata.org/smallpox' [Online Resource]BibTeX citation
@article{owid-smallpox,
author = {Sophie Ochmann and Saloni Dattani and Max Roser},
title = {Smallpox},
journal = {Our World in Data},
year = {2018},
note = {https://ourworldindata.org/smallpox}
}Reuse this work freely
All visualizations, data, and code produced by Our World in Data are completely open access under the Creative Commons BY license. You have the permission to use, distribute, and reproduce these in any medium, provided the source and authors are credited.
The data produced by third parties and made available by Our World in Data is subject to the license terms from the original third-party authors. We will always indicate the original source of the data in our documentation, so you should always check the license of any such third-party data before use and redistribution.
All of our charts can be embedded in any site.