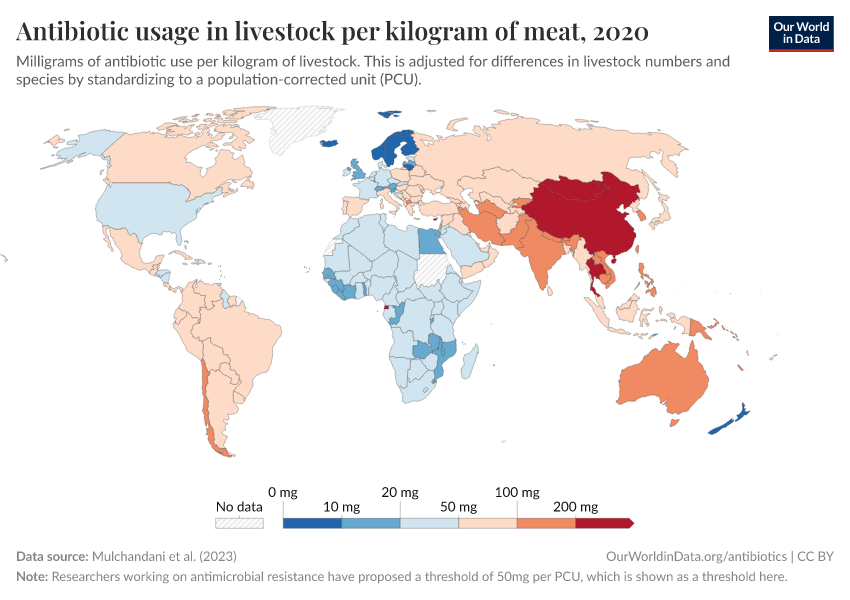
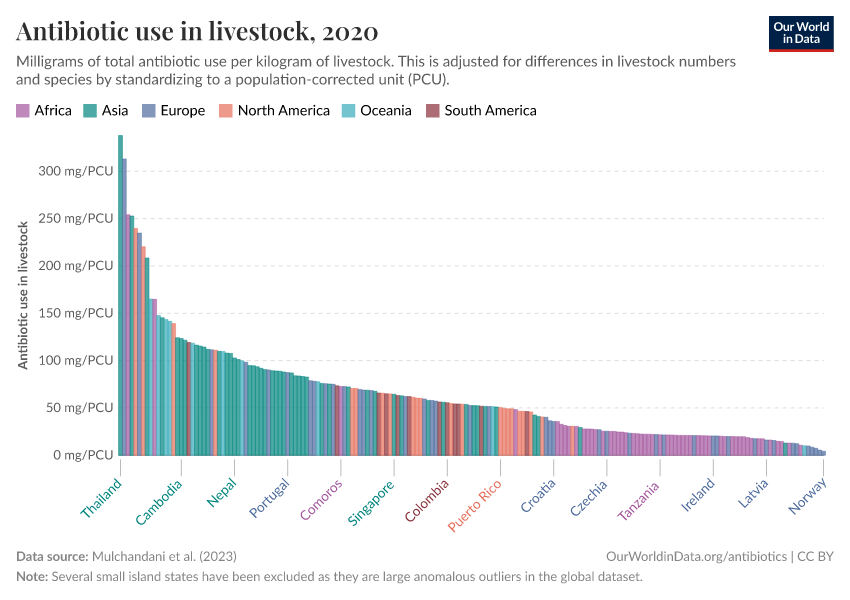
About this data
Related research and writing
Sources and processing
This data is based on the following sources
How we process data at Our World in Data
All data and visualizations on Our World in Data rely on data sourced from one or several original data providers. Preparing this original data involves several processing steps. Depending on the data, this can include standardizing country names and world region definitions, converting units, calculating derived indicators such as per capita measures, as well as adding or adapting metadata such as the name or the description given to an indicator.
At the link below you can find a detailed description of the structure of our data pipeline, including links to all the code used to prepare data across Our World in Data.
Reuse this work
Citations
How to cite this page
To cite this page overall, including any descriptions, FAQs or explanations of the data authored by Our World in Data, please use the following citation:
“Data Page: Antibiotic usage in livestock per kilogram of meat”. Our World in Data (2026). Data adapted from Mulchandani et al.. Retrieved from https://archive.ourworldindata.org/20260518-093348/grapher/antibiotic-usage-in-livestock.html [online resource] (archived on May 18, 2026).How to cite this data
In-line citationIf you have limited space (e.g. in data visualizations), you can use this abbreviated in-line citation:
Mulchandani et al. (2023) – processed by Our World in DataFull citation
Mulchandani et al. (2023) – processed by Our World in Data. “Antibiotic usage in livestock per kilogram of meat” [dataset]. Mulchandani et al., “Global trends in antimicrobial use in food-producing animals: 2020 to 2030” [original data]. Retrieved June 7, 2026 from https://archive.ourworldindata.org/20260518-093348/grapher/antibiotic-usage-in-livestock.html (archived on May 18, 2026).Download
Quick download
Download the data shown in this chart as a ZIP file containing a CSV file, metadata in JSON format, and a README. The CSV file can be opened in Excel, Google Sheets, and other data analysis tools.
Data API
Use these URLs to programmatically access this chart's data and configure your requests with the options below. Our documentation provides more information on how to use the API, and you can find a few code examples below.
Data URL (CSV format)
https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.csv?v=1&csvType=full&useColumnShortNames=falseMetadata URL (JSON format)
https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.metadata.json?v=1&csvType=full&useColumnShortNames=falseExcel / Google Sheets
=IMPORTDATA("https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.csv?v=1&csvType=full&useColumnShortNames=false")Python with Pandas
import pandas as pd
import requests
# Fetch the data.
df = pd.read_csv("https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.csv?v=1&csvType=full&useColumnShortNames=false", storage_options = {'User-Agent': 'Our World In Data data fetch/1.0'})
# Fetch the metadata
metadata = requests.get("https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.metadata.json?v=1&csvType=full&useColumnShortNames=false").json()R
library(jsonlite)
# Fetch the data
df <- read.csv("https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.csv?v=1&csvType=full&useColumnShortNames=false")
# Fetch the metadata
metadata <- fromJSON("https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.metadata.json?v=1&csvType=full&useColumnShortNames=false")Stata
import delimited "https://ourworldindata.org/grapher/antibiotic-usage-in-livestock.csv?v=1&csvType=full&useColumnShortNames=false", encoding("utf-8") clear