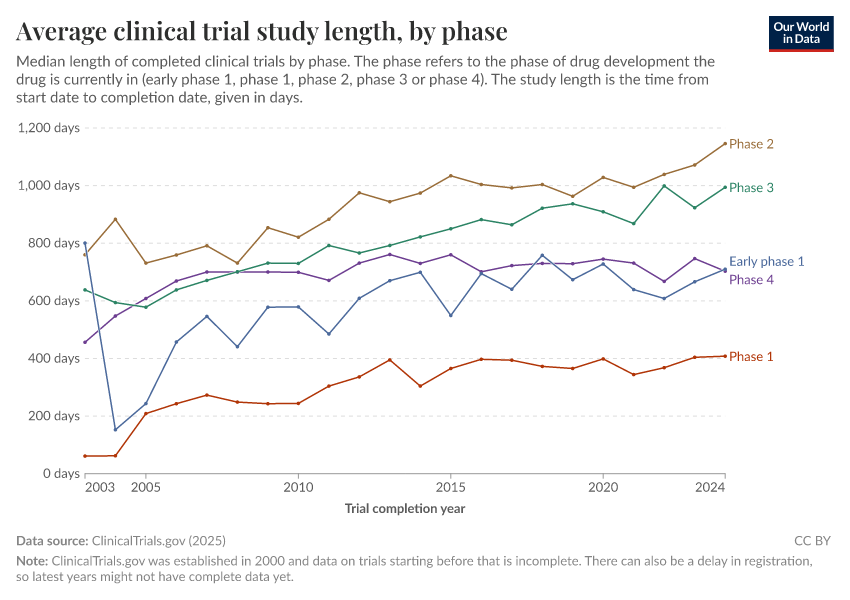
Average clinical trial study length, by phase
What you should know about this indicator
- Clinical trials are conducted in different phases of the development of a new treatment or intervention, such as early phase 1, phase 1, phase 2, phase 3, or phase 4.
- Early phase 1 trials are exploratory trials that test the effect of an intervention on the human body, often with a small number of participants. Not every treatment has an early phase 1 trial.
- Phase 1 trials test the safety and dosage of a new treatment, phase 2 trials test the effectiveness and side effects, and phase 3 trials compare the new treatment to standard treatments.
- Phase 4 trials are conducted after a treatment has been approved and are used to monitor the long-term effects and side-effects of the treatment. Not every treatment has a phase 4 trial.
- "Phase 1/phase 2" trials are a combination of phase 1 and phase 2 trials, and "phase 2/phase 3" trials are a combination of phase 2 and phase 3 trials.
- The average length is calculated by taking the difference between the start date and completion date of each trial, grouped by phase and completion year.
- This data comes from the ClinicalTrials.gov database. It only includes interventional clinical trials that are marked as "completed" and have a valid "completion date".
- Registration in the ClinicalTrials.gov database is mandatory for trials in the United States and for treatments that seek FDA approval, but voluntary for other trials conducted in other countries.
More Data on Medicine & Biotechnology
Sources and processing
This data is based on the following sources
How we process data at Our World in Data
All data and visualizations on Our World in Data rely on data sourced from one or several original data providers. Preparing this original data involves several processing steps. Depending on the data, this can include standardizing country names and world region definitions, converting units, calculating derived indicators such as per capita measures, as well as adding or adapting metadata such as the name or the description given to an indicator.
At the link below you can find a detailed description of the structure of our data pipeline, including links to all the code used to prepare data across Our World in Data.
Notes on our processing step for this indicator
- First we calculate the length of each trial by taking the difference between the start date and completion date of each trial.
- Then we group the trials by phase and completion year, and calculate the median length of each group.
Reuse this work
Citations
How to cite this page
To cite this page overall, including any descriptions, FAQs or explanations of the data authored by Our World in Data, please use the following citation:
“Data Page: Average clinical trial study length, by phase”, part of the following publication: Tuna Acisu, Saloni Dattani, Fiona Spooner, Veronika Samborska, Hannah Ritchie, and Max Roser (2025) - “Medicine and Biotechnology”. Data adapted from ClinicalTrials.gov. Retrieved from https://archive.ourworldindata.org/20260518-093348/grapher/average-study-length-by-phase.html [online resource] (archived on May 18, 2026).How to cite this data
In-line citationIf you have limited space (e.g. in data visualizations), you can use this abbreviated in-line citation:
ClinicalTrials.gov (2025) – with major processing by Our World in DataFull citation
ClinicalTrials.gov (2025) – with major processing by Our World in Data. “Average clinical trial study length, by phase” [dataset]. ClinicalTrials.gov, “Clinical Trials (ClinicalTrials.gov)” [original data]. Retrieved May 22, 2026 from https://archive.ourworldindata.org/20260518-093348/grapher/average-study-length-by-phase.html (archived on May 18, 2026).Download
Quick download
Download the data shown in this chart as a ZIP file containing a CSV file, metadata in JSON format, and a README. The CSV file can be opened in Excel, Google Sheets, and other data analysis tools.
Data API
Use these URLs to programmatically access this chart's data and configure your requests with the options below. Our documentation provides more information on how to use the API, and you can find a few code examples below.
Data URL (CSV format)
https://ourworldindata.org/grapher/average-study-length-by-phase.csv?v=1&csvType=full&useColumnShortNames=falseMetadata URL (JSON format)
https://ourworldindata.org/grapher/average-study-length-by-phase.metadata.json?v=1&csvType=full&useColumnShortNames=falseExcel / Google Sheets
=IMPORTDATA("https://ourworldindata.org/grapher/average-study-length-by-phase.csv?v=1&csvType=full&useColumnShortNames=false")Python with Pandas
import pandas as pd
import requests
# Fetch the data.
df = pd.read_csv("https://ourworldindata.org/grapher/average-study-length-by-phase.csv?v=1&csvType=full&useColumnShortNames=false", storage_options = {'User-Agent': 'Our World In Data data fetch/1.0'})
# Fetch the metadata
metadata = requests.get("https://ourworldindata.org/grapher/average-study-length-by-phase.metadata.json?v=1&csvType=full&useColumnShortNames=false").json()R
library(jsonlite)
# Fetch the data
df <- read.csv("https://ourworldindata.org/grapher/average-study-length-by-phase.csv?v=1&csvType=full&useColumnShortNames=false")
# Fetch the metadata
metadata <- fromJSON("https://ourworldindata.org/grapher/average-study-length-by-phase.metadata.json?v=1&csvType=full&useColumnShortNames=false")Stata
import delimited "https://ourworldindata.org/grapher/average-study-length-by-phase.csv?v=1&csvType=full&useColumnShortNames=false", encoding("utf-8") clear